Research Articles
Pre-Meal vs. Post-Meal Insulin: A Comprehensive Review of Clinical Protocols for Optimal Glycemic Control and Drug Development
This article provides a critical analysis for researchers and drug development professionals on the evolving clinical protocols for pre-meal (prandial) versus post-meal (postprandial) insulin administration.
Mastering Clarke Error Grid Analysis: Essential Validation Framework for Glucose Monitoring Systems in Biomedical Research
This comprehensive guide provides researchers, scientists, and drug development professionals with an authoritative framework for applying Clarke Error Grid Analysis (EGA) to glucose monitoring system validation.
Clarke Error Grid Analysis: The Essential Guide to Validating Glucose Prediction Models in Clinical Research
This comprehensive guide explores Clarke Error Grid Analysis (CEGA) as the gold-standard method for validating the clinical accuracy of glucose prediction models.
Validating LSTM Glucose Predictions: A Comprehensive Guide to Clark Error Grid Analysis for Biomedical Research
This article provides researchers, scientists, and drug development professionals with a comprehensive framework for validating Long Short-Term Memory (LSTM) models in biomedical applications, particularly glucose prediction.
Unpredictable Absorption: Decoding the Complex Drivers of Subcutaneous Insulin Variability in Drug Development
For researchers and drug development professionals, subcutaneous insulin absorption variability remains a critical challenge impacting efficacy, safety, and product development.
CamAPS FX Hybrid Closed-Loop System in Pregnancy: Comprehensive Clinical Trial Results and Analysis
This article provides a detailed analysis of recent clinical trial results for the CamAPS FX hybrid closed-loop (HCL) system in pregnant individuals with type 1 diabetes.
The Cahill Cycle (Alanine-Glucose Cycle): A Comprehensive Analysis of Physiology, Research Methods, and Therapeutic Potential
This review provides a detailed examination of the Cahill (alanine-glucose) cycle, a critical interorgan metabolic pathway essential for nitrogen transport and gluconeogenesis during fasting and exercise.
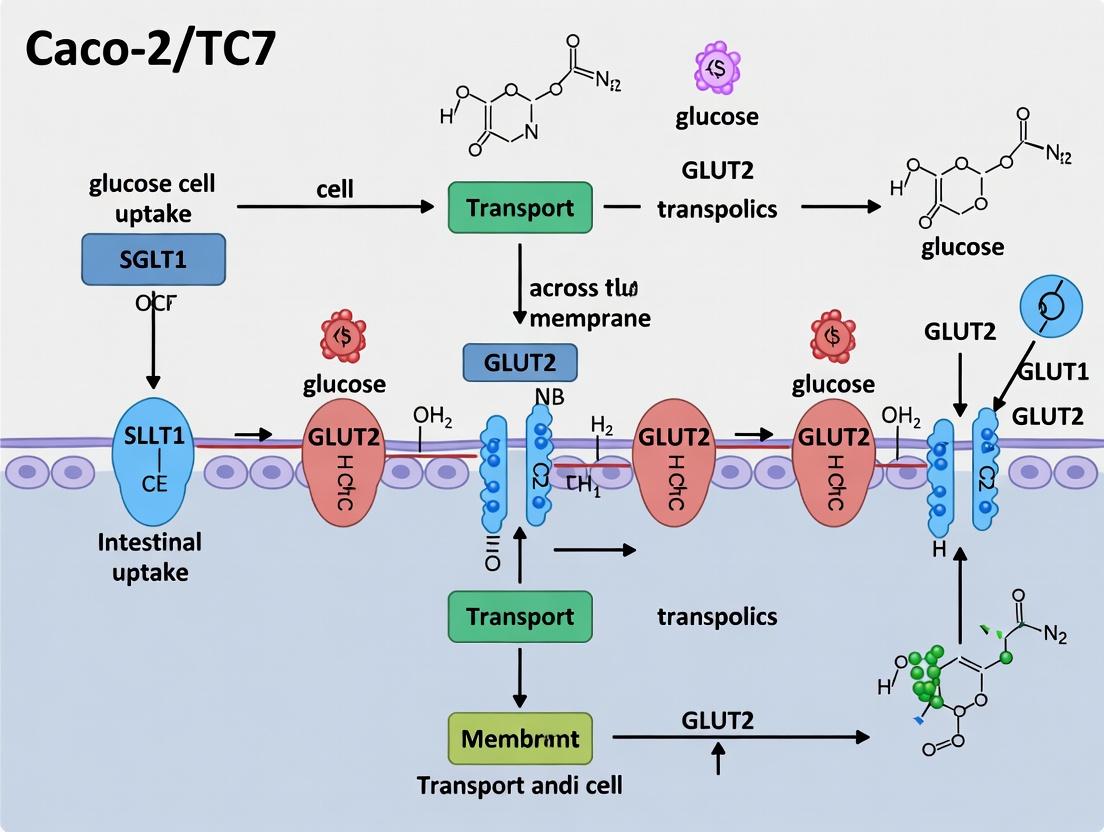
The Caco-2/TC7 Model: A Comprehensive Guide to Assessing Intestinal Glucose Uptake for Drug and Nutraceutical Research
This article provides researchers, scientists, and drug development professionals with a detailed, current guide to the Caco-2/TC7 intestinal epithelial cell model for glucose uptake assessment.
Mastering Caco-2 and TC7 Cell Models: A Complete Guide to Intestinal Glucose Transport Research
This comprehensive guide provides researchers and drug development professionals with an in-depth exploration of the Caco-2 and TC7 cell lines as premier in vitro models for studying intestinal glucose transport.
Caco-2 TC7 vs. Other Intestinal Models: A Researcher's Guide to Glucose Transport Studies
This comprehensive guide analyzes the Caco-2 TC7 clone in the context of glucose transport and permeability research.