The Hovorka Model in Closed-Loop Insulin Delivery: A Comprehensive Guide for Diabetes Research and Algorithm Development
This article provides a comprehensive technical examination of the Hovorka (Cambridge) model for closed-loop insulin delivery.
The Hovorka Model in Closed-Loop Insulin Delivery: A Comprehensive Guide for Diabetes Research and Algorithm Development
Abstract
This article provides a comprehensive technical examination of the Hovorka (Cambridge) model for closed-loop insulin delivery. Designed for researchers, scientists, and drug development professionals, it covers the mathematical foundations, physiological compartments, and key state variables of the model. We detail its methodological implementation in control algorithms, including real-time parameter estimation and MPC strategies. The guide addresses common challenges in parameter identification, model personalization, and handling inter-/intra-patient variability. Finally, we analyze clinical validation studies, benchmark the Hovorka model against other physiological models (e.g., UVa/Padova, Sorensen), and discuss its role in regulatory pathways for artificial pancreas systems. This resource synthesizes current knowledge to inform advanced algorithm design and translational research.
Deconstructing the Hovorka Model: Mathematical Foundations and Physiological Principles for Glucose-Insulin Dynamics
Origins and Philosophical Foundations
The Hovorka model, also known as the Cambridge model, is a physiological, compartmental model of glucose-insulin dynamics in individuals with Type 1 Diabetes (T1D). It was developed in the early 2000s by Professor Roman Hovorka and colleagues at the University of Cambridge. Its core philosophy is grounded in creating a mechanistic, patient-tailorable model to serve as a high-fidelity in-silico simulation environment for testing and developing closed-loop insulin delivery (artificial pancreas) systems. Unlike empirical "black-box" models, it explicitly represents key physiological subsystems—glucose absorption, insulin pharmacokinetics/pharmacodynamics, and endogenous glucose production—to facilitate individualized parameter estimation and credible long-term predictions.
Core Model Structure and Quantitative Parameters
The model is built around three interlinked subsystems. Its differential equations are parameterized using population and individual data.
Table 1: Core Subsystems and Key Parameters of the Hovorka Model
| Subsystem | Compartments | Key Parameters (Example Values) | Physiological Representation |
|---|---|---|---|
| Glucose Absorption | Gut (2 compartments) | ( D ) (meal glucose amount), ( k{12} ), ( k{a1} ) (rate constants) | Delayed appearance of meal-derived glucose into plasma. |
| Insulin Kinetics | Plasma, Effect (2 compartments) | ( k{aI} ) (absorption rate), ( t{max,I} ) (time-to-peak), ( V_I ) (distribution volume) | Subcutaneous insulin absorption and plasma clearance. |
| Glucose Kinetics | Plasma, Remote (2 compartments) | ( F{01c} ) (zero-order glucose excretion), ( EGP0 ) (endogenous production), ( S_{IT} ) (insulin sensitivity) | Glucose distribution, utilization, and insulin-dependent suppression of hepatic glucose production. |
Table 2: Typical Population Parameters (70kg Adult with T1D)
| Parameter | Symbol | Nominal Value | Unit |
|---|---|---|---|
| Insulin Sensitivity (Transport) | ( S_{IT} ) | 0.01 – 0.04 | L/min per mU |
| Insulin Sensitivity (Disposal) | ( S_{ID} ) | 0.01 – 0.03 | L/min per mU |
| Insulin Sensitivity (EGP) | ( S_{IE} ) | 0.0001 – 0.0003 | per mU |
| Glucose Effectiveness | ( EGP_0 ) | 1.0 – 1.5 | mmol/min |
| Distribution Volume (Glucose) | ( V_G ) | 0.16 | L/kg |
| Body Weight | ( BW ) | 70 | kg |
Experimental Protocols for Model Parameter Identification
For the model to be used in individualized control algorithms, its parameters must be estimated from subject data.
Protocol 1: Frequent-Sample Intravenous Glucose Tolerance Test (FSIGT) for Insulin Sensitivity Estimation
Objective: To estimate insulin sensitivity parameters ((S{IT}), (S{ID}), (S_{IE})) and glucose effectiveness. Materials: See Scientist's Toolkit. Procedure:
- Subject Preparation: Overnight fast (10-12 hrs). Insert IV cannulae in both arms (one for infusion, one for sampling).
- Baseline Sampling: At t = -10 and 0 min, collect plasma samples for glucose and insulin.
- Glucose Bolus: At t=0, rapidly inject 50% dextrose (0.3 g/kg body weight) over 1 minute.
- Frequent Sampling: Collect blood samples at 2, 3, 4, 5, 6, 8, 10, 12, 14, 16, 19, 22, 25, 30, 40, 50, 60, 70, 80, 90, 100, 120, 140, 160, and 180 minutes.
- Sample Analysis: Immediately centrifuge samples and analyze plasma glucose and insulin concentrations.
- Model Fitting: Use nonlinear least-squares software (e.g., SAAM II, MATLAB lsqnonlin) to fit the Hovorka model differential equations to the glucose and insulin time-series data, adjusting sensitivity parameters to minimize error.
Protocol 2: Meal Tolerance Test (MTT) for Glucose Absorption Model Calibration
Objective: To individualize meal glucose absorption parameters ((k{12}), (k{a1}), meal carbohydrate ratio). Procedure:
- Subject Preparation: Overnight fast. Insert IV cannula for sampling. Insert continuous glucose monitor (CGM).
- Baseline & Insulin: Measure fasting glucose. Administer standard pre-meal insulin bolus per subject's therapy.
- Standardized Meal: Consume a meal with precisely weighed carbohydrate content (e.g., 50g) within 15 minutes.
- Postprandial Monitoring: Sample blood for glucose and insulin at 15, 30, 60, 90, 120, 180, 240 mins. CGM records interstitial glucose continuously.
- Parameter Estimation: Fix insulin sensitivity parameters from FSIGT. Fit the glucose absorption submodel to the postprandial glucose excursion data, accounting for CGM time lag.
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for Hovorka Model In-Vivo Validation
| Item | Function/Description |
|---|---|
| Euglycemic-Hyperinsulinemic Clamp Apparatus | Gold-standard protocol to independently validate model-derived insulin sensitivity parameters. |
| Continuous Glucose Monitoring (CGM) System (e.g., Dexcom G6, Medtronic Guardian) | Provides high-frequency interstitial glucose data for model fitting and closed-loop algorithm input. |
| Insulin Pump (e.g., Dana Diabecare, Omnipod) | Programmable device for precise subcutaneous insulin delivery in experimental closed-loop trials. |
| Precise Meal Carbohydrate Kits | Standardized, weighed meals (liquid or solid) for reproducible glucose absorption challenges. |
| Automated Blood Sampler (e.g., Biostator legacy systems or custom systems) | Allows frequent plasma sampling with minimal distress, improving data density for model fitting. |
| Parameter Estimation Software (MATLAB with Optimization Toolbox, SAAM II, Monolix) | Platforms for solving differential equations and performing nonlinear mixed-effects modeling of population/individual data. |
Model Visualization and Workflow
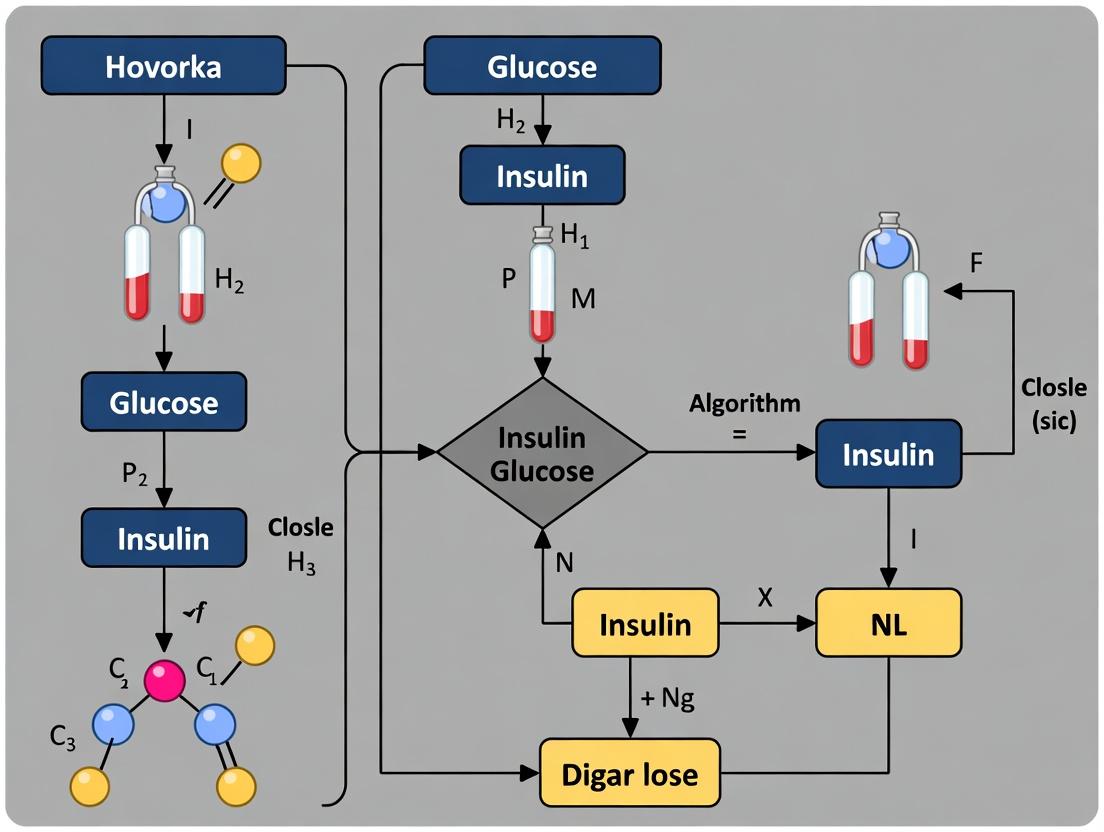
Diagram Title: Hovorka Model Parameterization and Control Loop
Diagram Title: Hovorka Model Core Compartmental Structure
This document details the mathematical framework of compartmental models, specifically within the context of a doctoral thesis researching next-generation closed-loop insulin delivery (artificial pancreas) control algorithms. The Hovorka model serves as the core physiological representation, and its refinement is critical for improving algorithm robustness and personalization. This framework provides the foundation for in silico testing, parameter identification, and control law derivation.
Core Compartmental Structure of the Hovorka Model
The Hovorka model is a nonlinear, deterministic model of glucose-insulin dynamics in Type 1 Diabetes. It consists of interconnected compartments representing subsystems.
Diagram 1: Hovorka Model Compartmental Overview
The model is defined by a set of coupled ordinary differential equations (ODEs). Below is a summary of the core state variables and their dynamics.
Table 1: Core State Variables and Differential Equations of the Hovorka Model
| Subsystem | State Variable (Unit) | Differential Equation (Key Terms) | Description |
|---|---|---|---|
| Glucose | G (mmol/L) | dG/dt = (Ra + EGP - E - Uii - ke1*G) / V_g | Plasma glucose concentration. V_g is distribution volume. |
| Insulin | I (mU/L) | dI/dt = -(ki1 + ki2)*I + S2 / V_i | Plasma insulin concentration. S2 is subcut. insulin infusion. |
| Insulin Action | x1, x2, x3 (1/min) | dxi/dt = -kai * x_i + k_ai * I (for i=1,2,3) | Insulin effects on EGP (x1), peripheral utilization (x2), and distribution (x3). |
| Subcutaneous Insulin | S1, S2 (mU) | dS1/dt = -ka1*S1 + IR; dS2/dt = ka1S1 - k_a2S2 | Two-compartment chain for delayed sc insulin absorption. IR is infusion rate. |
| Carbohydrates | D1, D2 (mmol) | dD1/dt = -kagD1 + CH; dD2/dt = k_a_gD1 - kag*D2 | Two-compartment chain for gut absorption of CHO (CH). |
Note: E = renal excretion, U_ii = insulin-independent utilization, k_xx are rate constants.
Application Notes: Parameter Identification Protocol
Experimental Protocol for Model Personalization
Aim: To identify individual-specific model parameters (e.g., insulin sensitivity, carbohydrate ratio) for controller tuning.
Materials & Reagent Solutions: Table 2: Research Reagent Solutions & Key Materials
| Item | Function/Description |
|---|---|
| Euglycemic-Hyperinsulinemic Clamp Setup | Gold-standard method for measuring insulin sensitivity. |
| Continuous Glucose Monitor (CGM) | Provides high-frequency interstitial glucose measurements. |
| Insulin Pump (programmable) | Delivers precise subcutaneous insulin infusion rates. |
| Reference Blood Glucose Analyzer (YSI) | Provides calibrated plasma glucose values for CGM alignment. |
| Standardized Meal Test Kits (e.g., Ensure) | Provides known carbohydrate load for absorption modeling. |
| Isotopic Glucose Tracer ([6,6-²H₂]glucose) | Allows precise measurement of endogenous glucose production (EGP). |
Parameter Estimation Software (e.g., MATLAB with fmincon, MONOLIX) |
Solves the inverse problem to fit model outputs to data. |
Procedure:
- Subject Preparation: Overnight fast (>8 hrs). Insert CGM and venous catheter for reference sampling.
- Basal Period (0-60 min): Collect reference glucose and insulin samples every 10-15 min to establish baseline.
- Meal Challenge (60-360 min): Administer standardized meal (e.g., 50g CHO). Record meal composition and time. Administer a patient-specific meal bolus via pump.
- Frequent Sampling: Collect reference glucose samples every 15-30 min. Record all insulin pump deliveries (basal, bolus).
- Data Preprocessing: Align CGM and reference data. Smooth CGM data with a low-pass filter if noise exceeds 10% MARD. Unit conversion for consistency.
- Parameter Estimation: a. Fixed Parameters: Set literature-based physiological constants (e.g., Vg, ka_i). b. Initial Guess: Use population median values for personalizable parameters (insulin sensitivity, carbohydrate ratio). c. Cost Function: Define as weighted sum of squared errors between model-predicted and measured glucose. d. Optimization: Execute constrained nonlinear optimization to minimize cost function, adjusting personalizable parameters.
- Validation: Simulate the personalized model against a separate dataset (e.g., a different meal day). Calculate root-mean-square error (RMSE) and correlation coefficient.
Diagram 2: Parameter Identification Workflow
Protocol: In Silico Stability Analysis of Control Algorithms
Detailed Methodology
Aim: To formally analyze the local stability of a closed-loop control law applied to the nonlinear Hovorka model.
Protocol Steps:
- Model Reduction: For analysis, reduce the full model to its core glucose-insulin-action feedback loops. A typical reduced model has states: G (glucose), I (insulin), x (insulin action).
- Control Law Definition: Define the controller (e.g., a Proportional-Integral-Derivative (PID) law operating on CGM error). The control output is the insulin infusion rate
IR(t).IR(t) = K_p * e(t) + K_i * ∫e(τ)dτ + K_d * (de/dt), wheree(t) = G_target - G_CGM(t). - Closed-Loop System: Form the augmented state-space by combining the reduced model ODEs with the controller's integral state ODE.
- Equilibrium Point Calculation: Solve for the steady-state (
x_ss) where all derivatives are zero under constant conditions (basal meal, no disturbance). - Jacobian Matrix Computation: Linearize the nonlinear closed-loop system around the equilibrium point
x_ss. Compute the Jacobian matrixJof partial derivatives.J_ij = ∂f_i/∂x_j |_(x=x_ss), wherefis the vector of ODEs. - Eigenvalue Analysis: Calculate the eigenvalues
λof the Jacobian matrixJ. - Stability Criterion: The equilibrium is locally asymptotically stable if and only if the real part of every eigenvalue is negative:
Re(λ_i) < 0for alli. - Sensitivity Analysis: Perform a parameter sweep of controller gains (
K_p, K_i, K_d). For each gain set, recompute eigenvalues. Map the region of stability in gain space.
Table 3: Sample Eigenvalue Analysis for a Given Gain Set
| Eigenvalue (λ) | Real Part | Imaginary Part | Stability Contribution |
|---|---|---|---|
| λ₁ | -0.0452 | +0.0000 | Stable (Negative Real) |
| λ₂ | -0.0211 | +0.0053 | Stable (Negative Real) |
| λ₃ | -0.0211 | -0.0053 | Stable (Negative Real) |
| λ₄ | -0.0015 | +0.0000 | Marginally Stable (Near Zero) |
Interpretation: The presence of an eigenvalue very close to zero indicates marginal stability, suggesting the need for controller retuning to improve robustness.
Application Notes and Protocols for Hovorka Model Research
This document provides detailed application notes and experimental protocols for investigating the key physiological compartment sub-models—glucose, insulin, and carbohydrate—that form the foundation of the Hovorka model. The Hovorka model is a widely used nonlinear differential equation model of glucose-insulin dynamics in type 1 diabetes, serving as a critical in-silico platform for developing and testing closed-loop insulin delivery (artificial pancreas) control algorithms. Understanding the mechanistic basis, parameterization, and validation of these core compartments is essential for advancing algorithm robustness, safety, and personalization.
Glucose Sub-model
The glucose sub-model describes the distribution and utilization of glucose in the body. It typically consists of two compartments: plasma glucose and glucose in the interstitial fluid/tissue space.
Table 1: Key States and Parameters of the Glucose Sub-model
| Symbol | Description | Typical Unit | Nominal Value (Example) | Physiological Meaning |
|---|---|---|---|---|
| G | Plasma glucose concentration | mmol/L | -- | State variable |
| Q1 | Glucose mass in accessible compartment (plasma) | mmol | -- | State variable |
| Q2 | Glucose mass in non-accessible compartment (tissue) | mmol | -- | State variable |
| VG | Distribution volume of glucose | L | 0.16 L/kg | Determines Q1 to G conversion |
| F01 | Insulin-independent glucose utilization | mmol/min | = 0.0037 * G (if G≥4.5) | Basal glucose consumption |
| EGP0 | Endogenous glucose production at zero insulin | mmol/min | 0.0161 mmol/kg/min | Hepatic glucose output |
| SIT | Insulin sensitivity of glucose disposal | L/min per mU | 0.001 ~ 0.02 | Governs insulin-mediated glucose uptake |
| SIE | Insulin sensitivity of endogenous glucose production suppression | L/mU | 0.0001 ~ 0.001 | Governs insulin's effect on liver |
Insulin Sub-model
This sub-model describes the pharmacokinetics of subcutaneously administered insulin, its absorption into plasma, and subsequent degradation.
Table 2: Key States and Parameters of the Insulin Sub-model
| Symbol | Description | Typical Unit | Nominal Value (Example) | Physiological Meaning |
|---|---|---|---|---|
| I | Plasma insulin concentration | mU/L | -- | State variable |
| S1, S2 | Insulin in subcutaneous compartments | mU | -- | States for delayed absorption |
| ka1, ka2 | Insulin absorption rate constants | min-1 | 0.006, 0.06 | Govern SC insulin absorption dynamics |
| ke | Insulin elimination rate constant | min-1 | 0.138 | Renal and peripheral degradation |
Carbohydrate Sub-model
This sub-model describes the appearance of glucose in the system from orally ingested carbohydrates, accounting for gut absorption delays.
Table 3: Key States and Parameters of the Carbohydrate Sub-model
| Symbol | Description | Typical Unit | Nominal Value (Example) | Physiological Meaning |
|---|---|---|---|---|
| D1, D2 | Glucose in gut compartments | mmol | -- | States for delayed absorption |
| kG | Carbohydrate absorption rate constant | min-1 | 0.05 ~ 0.07 | Governs rate of glucose entry from gut |
| AG | Bioavailable carbohydrate amount | g | -- | Input variable (meal) |
| BW | Body weight | kg | -- | Scaling factor |
Experimental Protocols for Model Parameter Identification
Protocol: Hyperinsulinemic-Euglycemic Clamp for Insulin Sensitivity Parameters
Objective: To quantify insulin sensitivity parameters (SIT, SIE) for individualizing the Hovorka model.
Materials: See "The Scientist's Toolkit" (Section 6). Procedure:
- Baseline Period (0-30 min): Insert intravenous cannulas for infusion and sampling. Measure fasting plasma glucose (G0) and insulin (I0).
- Priming & Infusion (0-120 min):
- Initiate a primed, continuous intravenous insulin infusion at a constant rate (e.g., 40 mU/m²/min).
- Simultaneously, begin a variable 20% dextrose infusion to maintain plasma glucose at the target euglycemic level (e.g., 5.0 mmol/L ± 0.5).
- Steady-State Period (90-120 min):
- The glucose infusion rate (GIR) is adjusted based on frequent plasma glucose measurements (every 5-10 min).
- Once GIR is stable for ≥30 minutes, the system is in steady state.
- Sampling & Analysis:
- During the steady-state period, collect plasma samples for precise glucose and insulin assay.
- Calculation: At steady state, the total glucose disposal rate (GRd) equals the GIR. Endogenous glucose production (EGP) is assumed fully suppressed.
- Insulin sensitivity index (M/I value) = GRd / (ΔI * BW), where ΔI is the steady-state increment in plasma insulin above baseline. This empirical index is used to scale model parameters SIT and SIE.
Protocol: Meal Tolerance Test for Carbohydrate Absorption Dynamics
Objective: To identify the carbohydrate absorption rate constant (kG) and meal bioavailability.
Procedure:
- Preparation: Participant fasts overnight (≥10 hrs). Insert a venous cannula for frequent sampling.
- Baseline Samples (t = -15, 0 min): Collect blood for glucose, insulin, C-peptide.
- Meal Ingestion (t = 0 min): Consume a standardized mixed meal (e.g., 50g available carbohydrate) within 15 minutes. Precisely record composition and weight.
- Postprandial Sampling: Collect blood samples at frequent intervals (e.g., 15, 30, 45, 60, 90, 120, 150, 180 min) for glucose and insulin measurement.
- Data Fitting: Using the Hovorka model with fixed insulin parameters, optimize kG and potentially a meal bioavailability factor to minimize the error between the simulated and measured plasma glucose trajectory. This is typically done via nonlinear least-squares fitting.
Visualization of Compartmental Structures & Relationships
Diagram 1: Hovorka Model Core Compartmental Structure (76 chars)
Diagram 2: Parameter Identification Workflow (44 chars)
The Scientist's Toolkit: Research Reagent Solutions
Table 4: Essential Materials for Hovorka Model Experimental Validation
| Item/Category | Example Product/Solution | Function in Research |
|---|---|---|
| Continuous Glucose Monitor (CGM) | Dexcom G7, Medtronic Guardian 4 | Provides high-frequency interstitial glucose data for model input and validation in ambulatory settings. |
| Insulin Pump | Dana Diabecare IIS, Omnipod DASH | Delivers precise subcutaneous insulin infusions as commanded by the control algorithm being tested. |
| Closed-Loop Control Platform | AndroidAPS, OpenAPS, Cambridge AP | Open-source or research-specific software that implements the Hovorka model and control algorithm for real-time testing. |
| Reference Blood Glucose Analyzer | YSI 2300 STAT Plus, Abbott Biosen C-line | Provides highly accurate plasma glucose measurements for calibrating CGM and validating model predictions (gold standard). |
| Human Insulin for Clamp | Actrapid, Humulin R | Used in hyperinsulinemic-euglycemic clamps to create a steady-state insulin level for sensitivity measurement. |
| Stable Isotope Tracers | [6,6-²H₂]Glucose, [U-¹³C]Glucose | Allows direct, model-based quantification of endogenous glucose production (EGP) and glucose rate of appearance (Ra) during experiments. |
| Parameter Estimation Software | MATLAB with SimBiology, R with FME/dMod packages |
Provides tools for nonlinear mixed-effects modeling and parameter optimization against experimental data. |
| In-Silico Simulation Environment | UVa/Padova T1D Simulator, Hovorka model implemented in Python | Enables safe, rapid, and reproducible testing of control algorithms before human trials. |
Within the broader thesis on the Hovorka model for closed-loop insulin delivery control algorithm research, the precise definition of state variables and parameters is foundational. The Hovorka model is a nonlinear, compartmental model of glucose-insulin dynamics in individuals with Type 1 Diabetes. This document provides a structured reference table of its core components and delineates protocols for their experimental determination, essential for validating and personalizing model-based control algorithms.
Critical State Variables and Model Parameters: Reference Tables
Table 1: Primary State Variables of the Hovorka Model
| Variable | Symbol | Unit | Physiological Interpretation | Compartment |
|---|---|---|---|---|
| Glucose in accessible compartment | ( G ) | mmol/L | Plasma glucose concentration | 1 (Glucose) |
| Insulin in accessible compartment | ( I ) | mU/L | Plasma insulin concentration | 2 (Insulin) |
| Insulin action on glucose distribution/transport | ( x_1 ) | 1/min | Delayed effect of insulin | 3 (Insulin Action) |
| Insulin action on glucose disposal | ( x_2 ) | 1/min | Delayed effect of insulin | 3 (Insulin Action) |
| Insulin action on endogenous glucose production | ( x_3 ) | 1/min | Delayed effect of insulin | 3 (Insulin Action) |
| Glucose in non-accessible compartment | ( Q_1 ) | mmol | Glucose in tissue fluid | 1 (Glucose) |
| Glucose in accessible compartment mass | ( Q_2 ) | mmol | Glucose in plasma and rapidly mixing fluids | 1 (Glucose) |
| Subcutaneous insulin depot 1 | ( S_1 ) | mU | Amount of insulin in first subcutaneous compartment | 4 (Subcutaneous Insulin) |
| Subcutaneous insulin depot 2 | ( S_2 ) | mU | Amount of insulin in second subcutaneous compartment | 4 (Subcutaneous Insulin) |
Table 2: Key Model Parameters of the Hovorka Model
| Parameter | Symbol | Unit | Typical Range | Description |
|---|---|---|---|---|
| Insulin sensitivity for disposal | ( S_{ID} ) | L/mU/min | 0.001 - 0.06 | Governs effect of insulin action ((x_2)) on glucose disposal. |
| Insulin sensitivity for production | ( S_{IE} ) | L/mU/min | 0.001 - 0.06 | Governs effect of insulin action ((x_3)) on endogenous glucose production. |
| Carbohydrate bioavailability | ( A_G ) | - | 0.5 - 0.9 | Fraction of ingested CHO that appears in the system. |
| Carbohydrate absorption rate | ( t_{maxG} ) | min | 20 - 80 | Time-to-max of CHO absorption. |
| Insulin absorption rate | ( t_{maxI} ) | min | 40 - 100 | Time-to-max of insulin absorption from subcutaneous tissue. |
| Time constant for insulin action | ( \tau ) | min | 40 - 120 | Governs the delay of insulin effect. |
| Endogenous glucose production at zero insulin | ( EGP_0 ) | mmol/min | 0.01 - 0.03 | Basal glucose production rate. |
| Glucose clearance at zero insulin | ( F_{01} ) | mmol/min | 0.01 - 0.02 | Basal glucose utilization. |
| Glucose distribution volume | ( V_G ) | L | 0.12 - 0.20 L/kg | Volume of the glucose distribution space. |
| Body weight | ( BW ) | kg | Individual | Used to scale several parameters. |
Experimental Protocols for Parameter Identification
Protocol 1: Hyperinsulinemic-Euglycemic Clamp for Estimating ( S{ID} ) and ( S{IE} )
Objective: To quantify insulin sensitivity parameters. Methodology:
- Preparation: Overnight fast. Insert intravenous catheters for insulin/glucose infusion and frequent blood sampling.
- Basal Period (0-120 min): Measure fasting plasma glucose and insulin.
- Insulin Infusion (120-360 min): Initiate a primed, continuous IV insulin infusion (e.g., 40 mU/m²/min) to achieve steady-state hyperinsulinemia.
- Glucose Clamping: Measure plasma glucose every 5 min. Adjust a variable 20% dextrose infusion rate to maintain euglycemia (e.g., 5.0 mmol/L).
- Steady-State Calculation: The last 30 min of the clamp define the steady state.
- Data Analysis: ( S{ID} ) (M-value) is calculated as the mean glucose infusion rate (GIR) normalized to body weight during steady-state. Model-based deconvolution of glucose and insulin traces yields estimates for ( S{ID} ) and ( S_{IE} ).
Protocol 2: Mixed-Meal Tolerance Test (MMTT) for Estimating ( AG ) and ( t{maxG} )
Objective: To characterize carbohydrate absorption dynamics. Methodology:
- Preparation: Overnight fast. Insert catheter for frequent sampling.
- Baseline Samples (-30, -15, 0 min): Collect blood for glucose, insulin, C-peptide.
- Meal Ingestion (0 min): Consume a standardized mixed meal (e.g., 75g CHO) within 15 minutes.
- Postprandial Sampling: Collect blood at 15, 30, 60, 90, 120, 180, 240 min for glucose, insulin, and optionally plasma acetaminophen (if co-ingested as a tracer for gastric emptying).
- Data Analysis: Use a deconvolution technique (e.g., Bayesian estimation) on the glucose appearance rate (derived from glucose and insulin models) to fit the two-compartment CHO absorption model, identifying ( AG ) and ( t{maxG} ).
Protocol 3: Subcutaneous Insulin Pharmacokinetic/Pharmacodynamic (PK/PD) Study for Estimating ( t_{maxI} ) and ( \tau )
Objective: To determine the absorption and action delay of subcutaneously administered insulin. Methodology:
- Preparation: Overnight fast, clamped at euglycemia via variable glucose infusion.
- Insulin Administration: Administer a bolus of rapid-acting insulin analog (0.1 U/kg) via subcutaneous injection.
- High-Frequency Sampling: Measure plasma insulin (or insulin analog concentration) and glucose every 10-20 min for 6-8 hours.
- Data Analysis: Fit a two-compartment insulin absorption model (e.g., ( S1, S2 )) to the plasma insulin concentration data to estimate ( t{maxI} ). Subsequently, fit the insulin action compartment model (( x1, x2, x3 )) to the glucose excursion (or glucose infusion rate needed to maintain euglycemia) to estimate the time constant ( \tau ).
Visualization of Model Structure and Experimental Workflow
Hovorka Model Compartmental Structure
Parameter Identification and Validation Workflow
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Research Materials and Reagents
| Item | Function in Research | Example/Notes |
|---|---|---|
| Human Insulin Analogs (IV/SC Grade) | Used in clamp studies and PK/PD protocols to induce controlled hyperinsulinemia. | Rapid-acting (Lispro, Aspart), Long-acting (Glargine, Detemir). Ensure high-purity, clinical grade. |
| 20% Dextrose Solution for Infusion | The exogenous glucose source for maintaining euglycemia during hyperinsulinemic clamps. | Must be sterile, pyrogen-free. Infusion rate is the primary clamp outcome measure (GIR). |
| Stable Isotope Glucose Tracers | Allows precise measurement of endogenous glucose production (EGP) and glucose rate of appearance (Ra) during mixed-meal studies. | [6,6-²H₂]-glucose; measured via GC-MS or LC-MS. |
| Acetaminophen (Paracetamol) | A marker for gastric emptying rate when co-ingested with a meal; informs the initial phase of CHO absorption. | Often given with the test meal; plasma concentration is measured. |
| Specific Insulin/Insulin Analog ELISA Kits | Critical for accurate measurement of low plasma insulin concentrations, especially distinguishing endogenous from exogenous insulin. | Should have high specificity for the insulin analog used (e.g., Lispro-specific assay). |
| Continuous Glucose Monitor (CGM) | Provides high-frequency interstitial glucose data for model validation and real-time algorithm testing. | Used in closed-loop studies. Key metrics: MARD (Mean Absolute Relative Difference). |
| Modeling & Estimation Software | Platform for implementing the Hovorka model, performing parameter identification, and simulating control algorithms. | MATLAB/Simulink, R, Python (SciPy, PINTS), SAAM II, NONMEM. |
The Role of the Hovorka Model in the Evolution of Artificial Pancreas (AP) Research
The Hovorka model, a compartmental model of glucose-insulin dynamics, has been a cornerstone in the development of closed-loop insulin delivery systems, known as the Artificial Pancreas (AP). Its primary role has been to serve as a robust, physiologically-relevant in silico simulation environment for the design, testing, and validation of control algorithms before clinical trials.
Core Mathematical Framework & Quantitative Parameters
The model describes glucose kinetics in multiple compartments: plasma, rapidly-equilibrating tissues, and slowly-equilibrating tissues. Insulin action is partitioned into effects on glucose disposal, endogenous glucose production, and glucose transport.
Table 1: Core Parameters of the Standard Hovorka Model
| Parameter Symbol | Description | Typical Value (70kg Adult) | Units |
|---|---|---|---|
| F01 | Non-insulin-dependent glucose flux | 0.0097 | mmol/min |
| EGP0 | Endogenous glucose production at zero insulin | 0.0161 | mmol/min |
| SIT | Insulin sensitivity (transport/deceleration) | 0.005 | L/min per mU |
| SID | Insulin sensitivity (disposal) | 0.00004 | L/min per mU |
| SIE | Insulin sensitivity (endogenous production) | 0.0002 | L/min per mU |
| ka1, ka2, ka3 | Deactivation rate constants for insulin action | 0.006, 0.06, 0.03 | min⁻¹ |
| VG | Distribution volume for glucose | 0.16 | L/kg |
| k12 | Transfer rate from compartment 2 to 1 | 0.066 | min⁻¹ |
| tmax,I | Time-to-max of insulin absorption | 55 | min |
| Bio | Bioavailability of subcut. insulin | 0.8 | - |
Application Notes: From Simulation to Clinical Implementation
Note 1: In Silico Testing and the FDA Acceptance
The Hovorka model is integral to the University of Virginia/Padova (UVA/Padova) Type 1 Diabetes Simulator, accepted by the FDA as a substitute for pre-clinical animal trials for certain AP system components. It allows for the safe, rapid, and cost-effective testing of novel control algorithms (e.g., PID, MPC, Fuzzy Logic) across a virtual population with varying insulin sensitivities, meal sizes, and daily routines.
Note 2: Personalization and Adaptive Control
Modern AP research uses the model structure for real-time parameter estimation. By fitting model parameters to individual patient data (CGM, insulin delivery), algorithms can adapt to diurnal changes in insulin sensitivity, mitigating hyper- and hypoglycemic risks.
Note 3: Integration with Model Predictive Control (MPC)
The Hovorka model is frequently the internal "prediction engine" in MPC algorithms. It forecasts future glucose trajectories based on current state, announced meals, and proposed insulin infusion rates, enabling optimal, proactive control.
Experimental Protocols
Protocol 1: In Silico Validation of a Novel Control Algorithm
Aim: To evaluate the safety and efficacy of a new MPC algorithm using the Hovorka model within the UVA/Padova Simulator. Materials: See "Scientist's Toolkit" below. Method:
- Population Selection: Select the 10-adult "validation cohort" from the simulator.
- Scenario Definition: Define a 3-day simulation scenario with standard meals (45g, 70g, 60g CHO) at 8:00, 13:00, and 19:00. Introduce a 30g unannounced snack at 22:00 on day 2 and a 20% overnight basal insulin underdose.
- Algorithm Configuration: Implement the novel MPC algorithm with the Hovorka model as its internal predictor. Set safety constraints (e.g., target glucose: 110 mg/dL, max insulin: 5 U/h).
- Simulation Execution: Run closed-loop simulations for all virtual subjects.
- Outcome Analysis: Calculate primary endpoints: % Time in Range (TIR, 70-180 mg/dL), % Time Below Range (<70 mg/dL, <54 mg/dL), % Time Above Range (>180 mg/dL), and Mean Glucose.
Protocol 2: Clinical Personalization of Model Parameters
Aim: To individualize the Hovorka model parameters for a specific patient to improve MPC performance. Materials: CGM device, insulin pump, continuous glucose monitor. Method:
- Data Collection: Under clinical supervision, collect 5-7 days of patient data: CGM readings, logged meal carbohydrate counts, and insulin pump delivery history.
- Parameter Estimation: Use a Bayesian estimation or maximum likelihood framework. Fix structural parameters (e.g., VG, k12). Estimate key metabolic parameters (SIT, SID, SIE, EGP0) by minimizing the difference between model-predicted and measured CGM traces.
- Validation: Use the personalized model to predict glucose for a subsequent 24-hour withheld period. Compare predictions to actual CGM using Root Mean Square Error (RMSE).
- Implementation: Upload personalized parameters into the patient's AP MPC algorithm.
Visualizations
Title: Hovorka Model's Role in AP Development Workflow
Title: Hovorka Model in an MPC Control Loop
The Scientist's Toolkit: Key Research Reagents & Materials
Table 2: Essential Resources for AP Research Using the Hovorka Model
| Item | Function & Application in Research |
|---|---|
| UVA/Padova T1D Simulator | Accredited in silico platform containing the Hovorka model for pre-clinical testing of AP algorithms. |
| CGM Data Stream (e.g., Dexcom G6, Medtronic Guardian) | Provides real-time or retrospective interstitial glucose measurements for model personalization and validation. |
| Insulin Pump Data | Historical or real-time bolus/basal data essential for parameter estimation and simulating closed-loop delivery. |
| Parameter Estimation Software (e.g., MATLAB with fmincon, RStan) | Tools to fit the Hovorka model to individual patient data, optimizing metabolic parameters. |
| MPC Design Framework (e.g., ACADO, CasADi, YALMIP) | Software toolkits for implementing model predictive control using the Hovorka model as constraints. |
| Clinical Dataset (e.g., OhioT1D, Tidepool) | Open-source or proprietary datasets containing paired CGM, insulin, and meal data for model training and benchmarking. |
In the context of closed-loop insulin delivery (artificial pancreas) research, physiological models serve distinct, complementary purposes. The ecosystem ranges from high-level, control-oriented models to highly granular, mechanistic simulations. The Hovorka (Cambridge) model is a pivotal mid-fidelity model balancing physiological plausibility and computational efficiency for real-time control.
Comparative Taxonomy of Key Glucose-Insulin Models
The table below categorizes prominent models based on key characteristics relevant to control algorithm development.
Table 1: Comparative Analysis of Selected Physiological Models for Glucose-Insulin Dynamics
| Model Name (Primary Reference) | Core Purpose / Origin | Compartmental Structure (Glucose/Insulin) | Key Differentiating Features | Primary Application in Research | Suitability for Real-Time Control |
|---|---|---|---|---|---|
| Minimal Model (Bergman) | IVGTT analysis; theoretical foundation | 2 (Glucose) / 1 (Insulin) | "Minimal" identifiable parameters (SI, SG); gold standard for insulin sensitivity measurement. | Metabolic phenotyping, clinical assessment. | Low. Not designed for meal disturbance or closed-loop control. |
| Hovorka (Cambridge) Model | AP algorithm design & in silico testing | 3 (Glucose) / 2 (Insulin) | Comprehensive subcutaneous insulin absorption & glucose kinetics; accounts for insulin action on transport, disposal, and endogenous production. | In silico trials, MPC algorithm development, safety testing. | High. Designed explicitly for subcutaneous CGM/insulin pump AP systems. |
| UVa/Padova Simulator (FDA Accepted) | Pre-clinical in silico testing of AP algorithms | 13 (Non-linear) / 2 (Insulin) | A population of 100+ virtual "subjects" with inter-/intra-variability; FDA-recognized as a substitute for animal trials. | Benchmark validation of control algorithms pre-clinical trials. | Medium-High. Used for testing controllers, not typically embedded in the controller. |
| DMMS (Dual-Hormone Model) | Multi-hormone (Glucagon) AP research | Extends Hovorka/UVa models | Incorporates glucagon kinetics and action, enabling bi-hormonal control strategies. | Research on hypoglycemia mitigation and dual-hormone AP systems. | Medium (increased complexity). |
| Sorensen Model | Whole-body physiological simulation | 6 (Glucose) / 3 (Insulin) | Highly detailed organ-level compartmentalization (brain, heart, liver, gut, kidney, periphery). | Deep physiological investigation, educational tool. | Low. Computationally intensive, over-parameterized for control. |
Application Notes: The Hovorka Model in AP Research
Role in the Development Pipeline
The Hovorka model functions at two critical stages:
- Embedded in Model Predictive Control (MPC): Its state-space formulation allows it to be used directly within an MPC to predict future glucose trajectories and optimize insulin dosing.
- In Silico Evaluation (with UVa/Padova T1DMS): While the UVa/Padova simulator is the formal validation tool, the Hovorka model is often used for rapid, initial prototyping and tuning of controllers due to its faster simulation times and adequate realism.
Core Mathematical Structure & Parameters
The model describes a person with Type 1 Diabetes. Its key subsystems are:
- Subcutaneous Insulin Absorption: A two-compartment chain modeling the slow diffusion of rapid-acting insulin analogues from the injection site.
- Glucose Kinetics: A two-compartment model (accessible and non-accessible pools).
- Insulin Action: Three remote compartments representing insulin's effect on glucose disposal ((x1)), endogenous glucose production ((x2)), and, in some versions, transport ((x_3)).
- Endogenous Glucose Production (EGP) & Renal Excretion: EGP is suppressed by insulin and elevated by glucagon; glucose is excreted renally above a threshold.
Table 2: Key State Variables and Parameters of the Hovorka Model
| Symbol | Description | Typical Unit | Identifiable from Clinical Data? |
|---|---|---|---|
| (G) | Plasma glucose concentration | mmol/L | Yes (via CGM, with calibration) |
| (S1, S2) | Insulin in subcutaneous compartments | pmol/L | Indirectly (from insulin pump records) |
| (I) | Plasma insulin concentration | mU/L | No (rarely measured) |
| (x1, x2, x_3) | Insulin action compartments | 1/min | No (aggregated effect) |
| (F_{01}) | Non-insulin-dependent glucose flux | mmol/min | Population average |
| (S_{IT}) | Insulin sensitivity (disposal) | L/min per mU | Yes, critical for personalization |
| (S_{IE}) | Insulin sensitivity (EGP) | L/min per mU | Yes, critical for personalization |
| (t_{max,I}) | Time-to-max insulin absorption | min | Population/Formulation specific |
| (V_G) | Glucose distribution volume | L/kg | Population average |
Experimental Protocols
Protocol: Personalization of Hovorka Model Parameters from Clinical Data
Objective: To estimate patient-specific parameters ((S{IT}, S{IE}), possibly (t_{max,I})) for embedding in a personalized MPC algorithm. Materials: See "Research Reagent Solutions" below. Procedure:
- Data Collection Phase: Conduct a 24-48 hour observation period with the subject wearing a research-grade CGM and an insulin pump.
- Record all insulin boluses and basal rates.
- Record meal announcements (time, carbohydrate estimate).
- Record exercise sessions (start/end time, type, perceived intensity).
- Data Preprocessing:
- Align all time series (CGM, insulin, carbs) to a common 5-minute interval.
- Smooth CGM data using a low-pass filter (e.g., moving median) to reduce high-frequency noise without delaying the signal.
- Annotate periods of known sensor anomalies or missing data.
- Parameter Estimation (Nonlinear Optimization):
- Model: Use the Hovorka model ODEs as the forward simulator.
- Cost Function: Minimize the Root Mean Square Error (RMSE) between simulated plasma glucose ((G)) and measured CGM values, weighted by confidence in CGM readings.
- Algorithm: Employ a gradient-based (e.g., Levenberg-Marquardt) or population-based (e.g., Particle Swarm) optimizer.
- Constraints: Impose physiologically plausible bounds on parameters (e.g., (S_{IT} > 0)).
- Execution: Run the optimization over the first 18 hours of data.
- Validation:
- Simulate the model with the optimized parameters for the remaining 6 hours of data not used in estimation.
- Calculate validation metrics: RMSE, Mean Absolute Relative Difference (MARD), and time in range (70-180 mg/dL) concordance.
Protocol: In Silico Closed-Loop Trial Using the Hovorka Model
Objective: To test the performance and safety of a novel MPC algorithm before human trials. Materials: MATLAB/Simulink or Python with SciPy; Hovorka model code; meal challenge scenario library. Procedure:
- Virtual Cohort Definition: Define a population of 10-100 in silico "subjects" by drawing parameters ((S{IT}, S{IE}, V_G, etc.)) from log-normal distributions fitted to real population data.
- Controller Integration: Implement the MPC algorithm. At each 5-minute control step:
- The MPC receives the current CGM value (from the Hovorka model simulation, with added realistic noise).
- The MPC uses an instance of the Hovorka model (the prediction model) to forecast glucose over a 2-6 hour horizon.
- The optimizer computes the optimal basal insulin infusion trajectory.
- Scenario Testing: Simulate each virtual subject over 3-7 days under challenging conditions:
- Day 1: Standardized meals (30g, 60g, 80g CHO).
- Day 2: Missed meal bolus.
- Day 3: Post-prandial exercise.
- Incorporate realistic diurnal variation in insulin sensitivity.
- Outcome Analysis: Aggregate results across the cohort. Key Performance Indicators (KPIs):
- Primary: % Time in Range (TIR) 70-180 mg/dL.
- Safety: % Time <54 mg/dL (Level 2 hypoglycemia), % Time >250 mg/dL.
- Insulin Use: Total Daily Dose (TDD), bolus-to-basal ratio.
Visualizations
Diagram 1: Hovorka Model in the Model Ecosystem
Diagram 2: Hovorka Model Key Pathways
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for Hovorka Model-Based AP Research
| Item / Solution | Function in Research | Example / Specification Notes |
|---|---|---|
| Research-Grade CGM System | Provides high-frequency (e.g., every 5-min) glucose concentration data for model personalization and validation. | Dexcom G6 Pro, Abbott Libre Pro. Must allow raw data access with timestamps. |
| Programmable Insulin Pump | Delivers precise micro-boluses and records exact infusion history, critical for accurate simulation inputs. | Insulet Omnipod DASH (Research Mode), Tandem t:slim (Research Platform). |
| Parameter Estimation Software | Solves the inverse problem to fit model parameters to clinical data. | MATLAB fmincon or lsqnonlin, Python SciPy.optimize, or custom Particle Filter/PSO code. |
| In Silico Simulation Environment | Integrates the Hovorka model, control algorithm, and virtual subject/scenario for testing. | MATLAB/Simulink, Python with scipy.integrate.ode, Julia with DifferentialEquations.jl. |
| Virtual Population Database | Provides statistically realistic sets of Hovorka model parameters representing a T1D population. | Derived from public datasets (e.g., OhioT1DM) or generated from published distributions (Wilinska et al., 2010). |
| Meal & Exercise Challenge Library | Standardizes testing scenarios to enable fair comparison between control algorithms. | Includes carbohydrate amounts (30-100g), timing, and glycemic index profiles, plus aerobic exercise models. |
Implementing the Hovorka Model: From Simulation to Real-World Control Algorithm Design
This application note details the integration of the Hovorka model, a compartmental model of glucose-insulin dynamics, into a Model Predictive Control (MPC) framework. This integration is a core pillar of a broader thesis on developing a robust, personalized closed-loop insulin delivery (artificial pancreas) control algorithm. The Hovorka model's non-linear, physiologically-relevant structure makes it a powerful, albeit complex, candidate for in-silico testing and controller design within an MPC paradigm.
Key Components and Mathematical Foundation
The Hovorka model describes glucose kinetics across several compartments. Key differential equations are summarized below. The state-space representation is essential for MPC implementation.
Core Model Equations (Abridged):
- Glucose Subsystem: ( \frac{dG}{dt} = F{01}^{c} + x1(t)G + EGP0(1 - x3(t)) - U_{ii} - E )
- Insulin Action Subsystems: ( \frac{dxi}{dt} = -k{ai}xi(t) + k{ai}Si Ip(t) ) for (i=1,2,3)
- Insulin Pharmacokinetics: ( \frac{dI1}{dt} = -(k{a1} + k{d})I1(t) + u{ex}(t) ), ( \frac{dIp}{dt} = -(k{a2} + k{d})Ip(t) + k{d}I_1(t) )
Where (G) is plasma glucose, (x1, x2, x3) are insulin action states, (Ip) is plasma insulin, and (u_{ex}) is exogenous insulin infusion rate.
Model Parameterization Table
For MPC, parameters must be individualized or drawn from population studies.
Table 1: Key Hovorka Model Parameters for a Representative Adult (70 kg)
| Parameter | Description | Nominal Value | Unit |
|---|---|---|---|
| (S_{IT}) | Insulin sensitivity (transport) | (51.2 \times 10^{-4}) | L/mU/min |
| (S_{ID}) | Insulin sensitivity (disposal) | (8.2 \times 10^{-4}) | 1/min |
| (S_{IE}) | Insulin sensitivity (EGP) | (520 \times 10^{-4}) | L/mU/min |
| (EGP_0) | Endogenous glucose production at zero insulin | (16.7) | μmol/kg/min |
| (k_{a1}) | Insulin absorption rate (subcutaneous) | (0.006) | 1/min |
| (k_{a2}) | Insulin absorption rate (plasma) | (0.06) | 1/min |
| (k_{a3}) | Delay of insulin action on EGP | (0.03) | 1/min |
| (V_G) | Distribution volume for glucose | (0.16) | L/kg |
| (F_{01}^{c}) | Constant glucose utilization | (1.0) | μmol/kg/min |
Experimental Protocols for Model Validation & Controller Tuning
Protocol 3.1: In-Silico Simulation for Controller Benchmarking
- Objective: To evaluate the performance of the Hovorka-MPC controller against a standard PID controller under meal challenge scenarios.
- Methodology:
- Simulation Environment: Utilize the FDA-accepted UVA/Padova T1D Simulator (or a custom Simulink/Julia/Python environment) with the Hovorka model as the "plant" (virtual patient).
- Virtual Cohort: Define a cohort of 10 in-silico adult subjects with parameters varied ±30% from nominal (Table 1) to represent inter-individual variability.
- Control Algorithms: Implement Hovorka-MPC and a tuned PID controller in parallel.
- Scenario: Simulate a 24-hour period with three unannounced meals (45g, 60g, 70g CHO). Add ±20% uncertainty to meal size in the MPC's observer.
- Metrics: Record % Time in Range (TIR: 70-180 mg/dL), Time Below Range (TBR: <70 mg/dL), and Total Insulin Delivered.
- Expected Outcome: Hovorka-MPC should achieve >75% TIR with lower TBR compared to PID.
Protocol 3.2: Parameter Identification from Clinical Data
- Objective: To individualize Hovorka model parameters ((S{IT}, S{ID}, S_{IE})) using Continuous Glucose Monitor (CGM) and insulin pump data.
- Methodology:
- Data Collection: Obtain 5-7 days of blinded CGM data and logged insulin bolus/basal data from a subject.
- Meal Annotation: Accurately log meal carbohydrate estimates.
- Optimization Routine: Use a non-linear least squares (e.g., Levenberg-Marquardt) or Bayesian estimation algorithm to fit model-predicted glucose to CGM traces.
- Validation: Use a separate 2-day data segment not used for fitting to validate model prediction accuracy (Root Mean Square Error, RMSE).
- Deliverable: A personalized parameter set for integration into the subject-specific MPC.
Integration Workflow: Hovorka Model into MPC
The integration follows a sequential workflow from model preparation to closed-loop control.
Title: Hovorka MPC Integration Workflow
MPC Formulation with Hovorka Model
The core of the integration is the translation of the model into a receding-horizon optimization problem.
Cost Function and Constraints
The MPC solves the following problem at each sampling time (e.g., every 5 minutes):
[ \min{\Delta u} \sum{j=1}^{Np} || y{t+j|t} - r{t+j} ||^2Q + \sum{j=0}^{Nc-1} || \Delta u{t+j|t} ||^2R ] subject to: [ x{k+1} = f(xk, uk, dk) \quad \text{(Hovorka model dynamics)} ] [ u{min} \leq uk \leq u{max} \quad \text{(Insulin pump limits)} ] [ \Delta u{min} \leq \Delta uk \leq \Delta u{max} \quad \text{(Infusion rate change limits)} ]
Table 2: Typical MPC Tuning Parameters for Hovorka Model
| Parameter | Symbol | Typical Value | Role in Control |
|---|---|---|---|
| Prediction Horizon | (N_p) | 6 - 12 steps (30-60 min) | Length of future predictions. |
| Control Horizon | (N_c) | 2 - 4 steps | Degrees of freedom for optimization. |
| Glucose Weight | (Q) | 1.0 - 10.0 | Penalizes deviation from setpoint (e.g., 110 mg/dL). |
| Insulin Change Weight | (R) | 50 - 500 | Penalizes aggressive insulin changes (safety). |
| Sampling Time | (T_s) | 5 min | Determines discrete model resolution. |
State Estimation and Disturbance Rejection
An Extended Kalman Filter (EKF) is typically used to estimate unmeasurable states (e.g., insulin action (x2), (x3)) and reject unmeasured meal disturbances ((d_k)).
Title: MPC-EKF Closed-Loop Control Structure
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Tools for Hovorka-MPC Research
| Item | Function in Research | Example/Detail |
|---|---|---|
| High-Fidelity T1D Simulator | Provides a safe, ethical platform for in-silico testing and benchmarking of controllers. | UVA/Padova Simulator (accepted by FDA), Cambridge Simulator. |
| Numerical Computing Environment | Used for model implementation, MPC optimization, and data analysis. | MATLAB/Simulink, Python (NumPy, SciPy, CasADi), Julia. |
| Quadratic Programming (QP) Solver | Solves the core optimization problem at each MPC step in real-time. | OSQP, qpOASES, FORCES Pro, MATLAB's quadprog. |
| Parameter Estimation Toolbox | Identifies personalized model parameters from clinical data. | MATLAB's System Identification Toolbox, PyMC3 (for Bayesian). |
| Continuous Glucose Monitor (CGM) Data | Real-world glucose traces for model validation and controller tuning. | Dexcom G6, Medtronic Guardian, Abbott Libre (interpolated). |
| Extended Kalman Filter (EKF) Codebase | Estimates unmeasurable model states and meal disturbances. | Custom code or toolbox implementations (e.g., MATLAB's EKF). |
| Clinical Protocol Design Software | Plans in-silico or clinical validation studies (meal challenges, exercise). | A dedicated tool for simulating realistic patient scenarios. |
Real-Time Parameter Estimation and Adaptive Filtering Techniques for the Hovorka Model
Application Notes
Within the broader thesis on developing a robust closed-loop insulin delivery (artificial pancreas) control algorithm, real-time parameter estimation and adaptive filtering are critical for personalizing the Hovorka model. This physiological model of glucose-insulin dynamics in type 1 diabetes (T1D) is structurally fixed, but its parameters vary significantly between individuals and within an individual over time due to lifestyle, physiology, and metabolic changes.
Core Challenges & Adaptive Solutions
A primary research challenge is the model's nonlinearity and the time-varying nature of key parameters, such as insulin sensitivity (S_I) and glucose effectiveness (S_G). Non-adaptive controllers using fixed model parameters can lead to suboptimal glycemic control. The integration of real-time estimation techniques allows the control algorithm to "learn" and adapt to the patient's current metabolic state.
Prominent Technical Approaches:
- Recursive Least Squares (RLS) & Forgetting Factors: Used for online estimation of slowly varying parameters from continuous glucose monitor (CGM) and insulin pump data. A forgetting factor discounts older data, allowing the algorithm to track temporal parameter drifts.
- Extended Kalman Filter (EKF): A widely adopted method for joint state and parameter estimation of the nonlinear Hovorka model. The EKF linearizes the model around the current estimate and provides a probabilistic framework to handle measurement noise.
- Unscented Kalman Filter (UKF): An alternative to EKF that avoids linearization by using a deterministic sampling approach, often providing better estimation performance for highly nonlinear systems.
- Bayesian Estimation: Methods like particle filters can handle non-Gaussian noise and multi-modal distributions, offering high fidelity at increased computational cost.
The successful implementation of these techniques moves the thesis from a theoretical simulation framework toward a clinically viable adaptive control system, capable of mitigating intra- and inter-patient variability.
Table 1: Performance Comparison of Estimation Algorithms in Simulation Studies
| Algorithm | Key Tuned Parameters | Estimated Variables | Performance Metric (RMSE) | Computational Load | Key Reference (Example) |
|---|---|---|---|---|---|
| Recursive Least Squares | Forgetting Factor (λ: 0.95-0.99) | S_I, S_G, EGP |
10-15 mg/dL | Low | Hovorka et al., 2004 |
| Extended Kalman Filter | Process & Measurement Noise Covariances (Q, R) | All States + S_I |
8-12 mg/dL | Medium | Bequette, 2013 |
| Unscented Kalman Filter | Scaling Parameters (α, β, κ), Noise Covariances | All States + S_I, S_G |
7-11 mg/dL | Medium-High | Vallis et al., 2020 |
| Particle Filter | Number of Particles (N: 500-2000), Proposal Distribution | All States + S_I, S_G |
6-10 mg/dL | High | Thabit & Hovorka, 2016 |
Table 2: Clinically Relevant Hovorka Model Parameters for Estimation
| Parameter Symbol | Description | Nominal Value (70kg) | Unit | Variability & Impact |
|---|---|---|---|---|
| S_I | Insulin Sensitivity | 5.0e-4 - 12.0e-4 | L/mU/min | High daily variability; primary adaptive target. |
| S_G | Glucose Effectiveness | 0.01 - 0.03 | 1/min | Modest variability; affects glucose disposal. |
| EGP₀ | Endogenous Glucose Production at zero insulin | 1.0 - 1.5 | mmol/min | Decreases with prolonged hyperglycemia. |
| F₀₁ | Bioavailability of injected insulin | 0.8 - 1.0 | Dimensionless | Can vary with injection site. |
| tmaxI | Time-to-maximum insulin absorption | 40 - 70 | min | Affects post-meal control. |
Experimental Protocols
Protocol 1: In Silico Validation Using the UVa/Padova T1D Simulator
Objective: To benchmark the performance of an EKF-based S_I estimator against a known "ground truth" in a controlled simulation environment.
- Platform: Use the accepted UVa/Padova T1D Simulator (version 2021 or later).
- Cohort: Select the 10-adult cohort. Simulate each virtual subject over a 3-day period with standardized meals (breakfast: 50g, lunch: 70g, dinner: 80g) and basal-bolus therapy.
- Intervention: Replace the simulator's native controller with the adaptive algorithm integrating the Hovorka model and the EKF.
- Estimation Setup: Initialize the EKF with population-average parameters. Set
S_Ias the only estimated parameter. Tune process noise (Q) forS_Ito reflect expected daily variation (~20-30%). - Data Streams: Feed the algorithm with simulated CGM data (5-minute intervals, add Gaussian noise, σ=2-3 mg/dL) and logged insulin delivery data.
- Validation: Record the estimated
S_Itrajectory. Compare to the simulator's internal, trueS_Iprofile (accessible via advanced logging). Calculate correlation and time-lag metrics. - Outcome Metrics: Primary: Time-in-Range (70-180 mg/dL). Secondary: Estimation RMSE for
S_I, computational time per step.
Protocol 2: Single-Hormone Closed-Loop Clinical Study with Adaptive Parameter Estimation
Objective: To evaluate the safety and efficacy of an adaptive closed-loop system using RLS for model personalization in a clinical research center.
- Participants: N=20 adults with T1D (HbA1c 7.0-9.5%), on insulin pump therapy.
- Study Design: Randomized, crossover trial comparing Adaptive Closed-Loop (ACL) vs. Non-Adaptive Closed-Loop (NACL) over two 36-hour inpatient periods.
- System Setup:
- ACL Arm: The control algorithm uses a Hovorka model with
S_IandS_Gupdated every 15 minutes using an RLS estimator (forgetting factor λ=0.98). Initial values are personalized from a pre-study basal titration. - NACL Arm: The algorithm uses the same Hovorka model but with static, population-derived parameters.
- ACL Arm: The control algorithm uses a Hovorka model with
- Procedure: Participants undergo identical meal challenges (e.g., 50g carbohydrate dinner) and overnight monitoring. Venous blood samples are drawn hourly for YSI glucose analyzer reference. Insulin delivery, CGM, and estimated parameters are logged.
- Safety & Analysis: A predefined hypoglycemia mitigation protocol is in place. Statistical analysis compares %Time-in-Range between arms using paired t-tests. Parameter estimates are analyzed for correlation with participant characteristics (e.g., total daily insulin dose).
Diagrams
Diagram Title: Adaptive Closed-Loop Control with RLS Estimation
Diagram Title: Clinical Trial Protocol for Adaptive Algorithm
The Scientist's Toolkit
Table 3: Key Research Reagent Solutions for Adaptive Hovorka Model Research
| Item Name | Provider/Example | Function in Research |
|---|---|---|
| UVa/Padova T1D Simulator | UVA Center for Diabetes Technology | The regulatory-accepted in-silico platform for closed-loop algorithm prototyping, validation, and benchmarking against a virtual cohort. |
| Continuous Glucose Monitor (Research Grade) | Dexcom G6 Pro, Abbott Libre Pro | Provides the essential, time-series glucose data stream (y_meas) for real-time parameter estimation. Research versions allow blinded data collection. |
| Insulin Pump (Research Interface) | Insulet Omnipod DASH Platform, Tandem t:slim X2 | Programmable pumps with communication APIs enable precise delivery logging (u) and automated control commands from a external algorithm. |
| Reference Blood Analyzer | YSI 2300 STAT Plus | Provides high-accuracy venous blood glucose measurements for calibrating CGM data and validating the accuracy of the overall system (ground truth). |
| Matlab/Simulink with Toolboxes | MathWorks | The dominant software environment for implementing the Hovorka model, designing estimation filters (EKF/UKF), and building Model Predictive Control (MPC). |
| Bayesian Estimation Toolbox | STAN, PyMC3 (Python) | Open-source probabilistic programming languages used for implementing advanced estimation methods like Particle Filters or Markov Chain Monte Carlo (MCMC). |
| Clinical Data Management System | OpenCDMS, REDCap | Securely manages and anonymizes patient data collected during clinical trials, including CGM traces, insulin logs, and parameter estimates. |
This Application Note details the critical process of designing a cost function for a closed-loop insulin delivery (CLID) control algorithm, specifically within the framework of a broader thesis on the Hovorka metabolic model. The Hovorka model is a differential-equation-based representation of glucose-insulin dynamics in individuals with Type 1 Diabetes (T1D). The efficacy of a Model Predictive Control (MPC) algorithm, a leading approach for CLID, is fundamentally determined by its cost function. This function mathematically encodes the clinical objectives: maintaining glucose within a safe target range, minimizing hypoglycemia risk, and delivering insulin in a safe, physiologically plausible manner. This document provides protocols for constructing, tuning, and validating this core component.
Core Components of the Cost Function
A typical cost function J for MPC in the Hovorka model context is a weighted sum of penalty terms over a prediction horizon (N steps). It balances glucose regulation against control effort and safety.
General Form:
J(k) = Σ_{i=1}^{N} [ w_g * (G(k+i|k) - G_target)^2 + w_Δu * (Δu(k+i-1))^2 + w_u * (u(k+i-1) - u_basal)^2 ] + Penalty_Terms
Quantitative Breakdown of Cost Function Terms
Table 1: Standard Cost Function Terms and Typical Weight Ranges
| Term | Mathematical Expression | Clinical/Algorithmic Purpose | Typical Weight Range (Relative) | Tuning Consideration |
|---|---|---|---|---|
| Glucose Deviation | w_g * (G - G_target)^2 |
Drives glucose towards the target setpoint (e.g., 110-120 mg/dL). | 1 (Reference) | Highest priority. Increased weight tightens control but may cause aggressiveness. |
| Insulin Change (Δu) | w_Δu * (Δu)^2 |
Penalizes rapid, large changes in insulin infusion rate. Promotes smoother delivery and actuator wear. | 10⁻² to 10⁻¹ | Critical for stability. Higher weight reduces oscillations but may slow response to meals. |
| Insulin Deviation | w_u * (u - u_basal)^2 |
Penalizes total insulin deviation from pre-programmed basal rate. Prevents over-dosing. | 10⁻³ to 10⁻² | Prevents "insulin stacking." Important for safety. |
| Hypoglycemia Penalty | Asymmetric quadratic or exponential function on low glucose. | Heavily penalizes predicted glucose values below a threshold (e.g., 80 mg/dL). | Function-specific | Non-linear. Must be severe enough to virtually forbid controller-induced lows. |
| Hyperglycemia Penalty | Asymmetric quadratic or linear function on high glucose. | Increases penalty for values above a higher threshold (e.g., 180 mg/dL). | Function-specific | Can be less aggressive than hypoglycemia penalty due to slower risks. |
Advanced Penalty Structures
Recent research incorporates more sophisticated terms:
- Zone-based Cost: Different weights for different glucose zones (hypo, target, hyper).
- Risk Index: Uses a published Blood Glucose Risk Index (BGRI) as a penalty term.
- Insulin-on-Board (IOB) Constraints: Penalizes dosing when significant active insulin is predicted.
Experimental Protocol: Cost Function Tuning & Validation
This protocol outlines a simulation-based methodology for tuning the penalty weights (w_g, w_Δu, w_u) and validating overall controller performance.
Protocol Title: In Silico Tuning and Validation of MPC Cost Function for Hovorka-Model CLID.
Objective: To systematically determine an optimal set of penalty weights that minimizes glycemic risk while ensuring safe insulin delivery profiles across a virtual patient cohort.
Materials & Reagent Solutions: Table 2: Research Reagent Solutions & Essential Materials
| Item | Function / Explanation |
|---|---|
| Hovorka Model Simulator | Core physiological model. Requires parameter sets for a diverse virtual cohort (e.g., adults, adolescents, varying insulin sensitivities). |
| Food & Meal Database | Standardized meal announcements (carbs, timing) with realistic variability for challenge scenarios. |
| Physical Activity Profile | Simulated or recorded heart rate/acceleration data to model exercise-induced glucose changes. |
| Insulin Pharmacokinetic/Pharmacodynamic (PK/PD) Model | Often embedded within Hovorka model. Describes subcutaneous insulin absorption and action. |
| Disturbance & Noise Model | Injects realistic CGM sensor noise, meal absorption uncertainty, and inter-day metabolic variability. |
| Performance Metric Calculator | Scripts to compute % Time in Range (70-180 mg/dL), Time Below Range (<70 mg/dL), Time Above Range (>180 mg/dL), Glucose Risk Index, and Total Insulin Dose. |
Methodology:
- Baseline Establishment: Simulate the virtual cohort under open-loop (basal insulin only) and standard therapy (basal-bolus) scenarios. Record all performance metrics.
- Weight Initialization: Define a broad search space for
[w_g, w_Δu, w_u]based on literature (see Table 1). Use logarithmic scaling. - Closed-Loop Simulation: For each weight combination: a. Initialize the MPC controller with the Hovorka model as the internal prediction model. b. Run a multi-day simulation for each virtual subject, incorporating announced meals, unannounced snacks, and simulated exercise sessions. c. Subject the controller to realistic challenges: sensor dropouts, pump occlusion alarms, and incorrect meal estimates.
- Primary Outcome Analysis: Calculate the mean and standard deviation of the performance metrics across the cohort for each weight set.
- Multi-Objective Optimization: Plot key outcomes against each other (e.g., % Time in Range vs. Time Below Range). Identify the Pareto front—the set of weight combinations where improving one metric worsens another.
- Safety-Focused Selection: From the Pareto-optimal sets, select the combination that achieves <1% Time Below Range (<70 mg/dL) as the highest priority, while maximizing Time in Range.
- Robustness Validation: Test the selected weight set on an untuned virtual cohort (new Hovorka model parameters not used during tuning) and under more extreme disturbance conditions.
Diagram: MPC Cost Function Tuning Workflow
Diagram Title: Workflow for tuning MPC cost function weights via simulation.
Visualization: Cost Function Logic in MPC
Diagram: Role of Cost Function in Hovorka-Model MPC
Diagram Title: Cost function's role in Hovorka-model MPC optimization cycle.
Designing the cost function is the central engineering challenge in translating the Hovorka model into a safe and effective clinical controller. The process involves a careful, simulation-driven trade-off between glycemic performance and insulin delivery safety, explicitly encoded through penalty weights. The protocols outlined here provide a rigorous, reproducible methodology for achieving this balance, forming a critical chapter in thesis research aimed at advancing closed-loop insulin delivery systems.
Within the broader thesis on the Hovorka model for closed-loop insulin delivery (artificial pancreas) research, a critical phase involves transitioning the validated control algorithm from simulation environments to real-world, embedded clinical systems. This application note details the principal practical implementation challenges—computational load, sampling time, and embedded system deployment—and provides experimental protocols for their systematic evaluation.
Core Challenges and Quantitative Analysis
Computational Load of the Hovorka Model
The Hovorka model, a system of nonlinear ordinary differential equations, presents a significant computational burden. The load is primarily dictated by the complexity of the model equations and the numerical integration method used by the Model Predictive Control (MPC) algorithm.
Table 1: Computational Load for Single Hovorka Model Prediction Horizon Evaluation
| Parameter | Value Range | Impact on Compute Time (ms) | Notes |
|---|---|---|---|
| States (ODEs) | 8 - 12 (core) | 5 - 15 | Depends on model variant (glucose, insulin, carbohydrates). |
| Prediction Horizon (Np) | 30 - 90 min | 20 - 200 | Linear increase with steps; major driver of load. |
| Control Horizon (Nc) | 1 - 3 steps | 5 - 50 | Affects optimization problem complexity. |
| Integration Step Size | 1 - 5 min | 2 - 10 | Smaller steps increase iterations. |
| Solver Type | Euler / RK4 | 1x / 3-5x | Runge-Kutta 4th order (RK4) is more accurate but heavier. |
| Platform | Desktop vs. ARM Cortex-M4 | 10x - 100x slowdown | Embedded processors lack FPU/advanced caches. |
Sampling Time Constraints
The sampling time ((T_s)) is the fixed interval at which the controller reads sensor data, executes the algorithm, and commands the insulin pump. It is bounded by the continuous glucose monitor (CGM) output rate and real-time requirements.
Table 2: Sampling Time Requirements and Implications
| System Component | Typical Rate/Constraint | Implementation Implication |
|---|---|---|
| CGM Data Output | 1 - 5 minutes | Defines the minimum possible (T_s). |
| Control Algorithm Execution | Must be < (T_s) | Total compute time must leave margin for I/O and safety checks. |
| Insulin Pump Communication | Per command | Adds fixed latency (~1-2s). |
| Hard Real-Time Deadline | (T_s) (e.g., 5 min) | Missing deadline is a critical system failure. |
| Recommended Margin | < 50% of (T_s) | Ensures robustness against timing jitter. |
Embedded Deployment Specifications
Deploying on a microcontroller unit (MCU) introduces constraints on memory, processing, and power.
Table 3: Embedded Platform Resource Allocation (Example: ARM Cortex-M4F @ 80MHz)
| Resource | Hovorka MPC Algorithm Usage | Typical MCU Limit | Utilization Risk |
|---|---|---|---|
| Flash/ROM | 50 - 150 kB | 512 kB - 1 MB | Low (includes firmware, RTOS). |
| RAM | 20 - 80 kB | 128 - 256 kB | Medium-High (stacks, matrices, buffers). |
| CPU Load per (T_s) | 2000 - 8000 ms | Must be < (T_s) (e.g., 300,000 ms @ 5 min) | Critical (Requires optimization). |
| FPU (Floating-Point Unit) | Mandatory | HW FPU (Cortex-M4F) | High (Software emulation is too slow). |
| Power Draw (Active) | 20 - 50 mA | Battery capacity driven (e.g., 500 mAh). | Medium (Impacts device lifespan). |
Experimental Protocols
Protocol: Profiling Computational Load on Target Hardware
Objective: To measure the worst-case execution time (WCET) of the control algorithm on the target embedded platform. Materials: See "The Scientist's Toolkit" (Section 5). Procedure: 1. Setup: Port the Hovorka MPC C-code to the target MCU (e.g., STM32F4). Enable a high-resolution hardware timer (e.g., SysTick). 2. Instrumentation: Insert timer start/stop calls at the beginning and end of the MPC calculation function. 3. Test Vectors: Generate a comprehensive set of input conditions (glucose history, insulin-on-board, meal announcements) covering physiological extremes. 4. Execution: Run the algorithm for each test vector for 1000 iterations. Record the execution time for each run. 5. Analysis: Calculate the maximum (WCET), minimum, average, and standard deviation of execution times. Ensure WCET is less than 50% of the intended sampling time (T_s).
Protocol: Closed-Loop Hardware-in-the-Loop (HIL) Testing with Fixed Sampling
Objective: To validate the integrated system's performance under realistic, fixed-time-step execution. Materials: MCU with deployed algorithm, HIL simulator (e.g., UVa/Padova T1D Simulator on a connected PC), real-time communication interface (UART/SPI). Procedure: 1. System Integration: Connect the MCU's I/O pins to the HIL simulator PC via a serial bridge. The MCU will receive "sensor" glucose and send "pump" commands. 2. Real-Time Scheduling: Implement a precise, interrupt-driven timer on the MCU to trigger the control cycle exactly every (Ts) (e.g., 300,000 ms). 3. Experiment Run: Initiate a 24-hour simulation scenario (including meals, exercise). The MCU algorithm runs in real-time, its execution time within each cycle logged. 4. Data Collection: Record glucose trajectories, insulin infusions, and most critically, any instances of *overrun* (where computation exceeds (Ts)). 5. Performance Metrics: Calculate % time in target range (70-180 mg/dL) for the HIL run and compare to non-real-time simulation results to quantify implementation penalty.
Diagrams
Diagram Title: Real-Time Control Cycle Logic Flow
Diagram Title: Hardware-in-the-Loop (HIL) Test Setup
The Scientist's Toolkit: Research Reagent Solutions
Table 4: Essential Tools for Implementation Challenge Research
| Item/Reagent | Function in Research | Example/Specification |
|---|---|---|
| Target MCU Development Board | Hardware platform for deployment and profiling. | STM32F407 Discovery (Cortex-M4F, 168 MHz, 192+ KB RAM). |
| Real-Time Operating System (RTOS) | Provides deterministic task scheduling for strict (T_s). | FreeRTOS, Zephyr OS. |
| Profiling Tools | Measure execution time and memory usage on embedded target. | Segger SystemView, ARM CMSIS-SVD viewer, GPIO toggling + Oscilloscope. |
| Hardware-in-the-Loop Simulator | Provides a realistic, reactive physiological environment for testing. | UVa/Padova T1D Simulator (FDA-accepted) with custom API for serial communication. |
| Static Code Analysis Tool | Ensures code safety, reliability, and efficiency pre-deployment. | MATLAB Polyspace, Klocwork, or Cppcheck for embedded C. |
| Fixed-Point Arithmetic Library | Optional tool to reduce compute load by replacing floating-point operations. | ARM CMSIS-DSP library, Qfmath (requires model linearization/validation). |
| Precision Timer Hardware | Enables microsecond-accurate timing for WCET measurement. | MCU's internal SysTick or a dedicated timer peripheral (e.g., TIM2). |
| Continuous Integration System | Automates build, test (including HIL), and profiling for each code change. | Jenkins or GitLab CI running cross-compilation and HIL test suites. |
Within the broader thesis on the Hovorka model for closed-loop insulin delivery, this review analyzes pivotal clinical trials that have translated the model's mathematical formalism into real-world therapeutic systems. The Hovorka model, a compartmental model of glucose-insulin dynamics, provides the physiological core for several advanced control algorithms. Its validation and refinement through these trials represent a critical pathway in the evolution of automated insulin delivery (AID) from research to clinical practice.
Review of Notable Closed-Loop Trials
The Florence Systems
A series of trials conducted by the University of Cambridge, culminating in the development of commercially available systems.
Key Trial: FlorenceD (2018)
- Objective: To evaluate the safety and efficacy of a closed-loop system using the Cambridge model predictive control (MPC) algorithm (rooted in the Hovorka model) in very young children with type 1 diabetes (T1D).
- Design: Randomized, crossover, outpatient trial.
- Population: Children aged 1–7 years.
- Intervention: 4 weeks of closed-loop (CamAPS FX app) vs. 4 weeks of sensor-augmented pump (SAP) therapy.
Table 1: Summary of Quantitative Outcomes from Key Florence-related Trials
| Trial (Year) | Population (n) | Design | Primary Outcome | Key Results (Closed-loop vs. Control) | Ref |
|---|---|---|---|---|---|
| FlorenceD (2018) | Children 1-7y (24) | RCT, crossover | % Time in Range (TIR, 3.9-10.0 mmol/L) | TIR: 74.6% vs 64.6% (Δ +10.0%, p=0.002). Time <3.9 mmol/L: 3.1% vs 3.7% (NS). | Lancet 2018 |
| CamAPS FX Pivotal (2020) | Adults & Children (136) | RCT, crossover | % TIR (3.9-10.0 mmol/L) | TIR: 65.3% vs 54.5% (Δ +10.8%, p<0.001). Mean glucose: 8.8 vs 9.5 mmol/L (p<0.001). | NEJM 2020 |
| FlorenceM (2020) | Pregnancy with T1D (16) | Open-label, single arm | % TIR (3.5-7.8 mmol/L) | Baseline TIR: 61%. Post-intervention TIR: 68% (p=0.028). No severe hypoglycemia. | Diabetologia 2020 |
CamAPS FX System Trials
The CamAPS FX hybrid closed-loop system is the commercial evolution of the Florence research platform, utilizing an Android smartphone app running a personalized MPC algorithm.
Key Trial: CamAPS FX Pivotal (2020)
- Objective: To assess the efficacy and safety of the CamAPS FX system in a broad population of adults, children, and adolescents with T1D.
- Design: Multicenter, randomized, crossover, outpatient trial.
- Population: Participants aged 6 years and older (n=136).
- Intervention: 4 months of CamAPS FX closed-loop vs. 4 months of insulin pump therapy (with or without continuous glucose monitoring [CGM]).
Detailed Experimental Protocols
Protocol 3.1: Outpatient Crossover Trial for AID System Evaluation (exemplified by CamAPS FX Pivotal Trial)
- Ethics & Recruitment: Obtain institutional review board approval. Recruit participants meeting inclusion criteria (e.g., diagnosis of T1D, age >6 years, use of insulin pump therapy).
- Randomization & Washout: Randomize participants to the order of intervention (closed-loop first or control first). No formal washout is typically used due to the crossover design; a 2–4 week run-in period on control therapy establishes baseline.
- Device Training: Provide comprehensive training on the study insulin pump, CGM, and the closed-loop system smartphone app (including carbohydrate counting and system alerts).
- Intervention Periods: Two 16-week treatment periods.
- Closed-loop: The MPC algorithm (Hovorka-based) runs on a locked smartphone. The system automatically adjusts basal insulin delivery every 8-12 minutes based on CGM readings. Users announce meals by entering carbohydrate estimates.
- Control: Continue usual insulin pump therapy (with CGM data blinded or open per standard care).
- Data Collection: CGM data is uploaded continuously. Participants maintain electronic diaries for meal announcements, insulin boluses, and exercise events. Safety data (severe hypoglycemia, diabetic ketoacidosis) are recorded.
- Endpoints Assessment: Primary outcome: % TIR (3.9–10.0 mmol/L) over the last 12 weeks of each period. Secondary outcomes: time above/below range, HbA1c, glucose variability, safety events, user-reported outcomes.
- Statistical Analysis: Use linear mixed models to compare outcomes between periods, accounting for period and carryover effects.
Protocol 3.2: In-Clinic Meal Challenge Sub-Study
- Objective: To intensively assess postprandial glucose control under closed-loop.
- Setting: Conducted within the main trial at selected centers on a subset of participants.
- Procedure: After an overnight fast and system initialization, participants are given a standardized, weighed mixed-meal (e.g., 60g carbohydrates). No pre-meal insulin bolus is administered by the participant; the closed-loop system responds autonomously. Frequent venous blood samples are taken for reference glucose measurement at intervals (e.g., -30, 0, 15, 30, 60, 90, 120, 180, 240 min).
- Analysis: Compare postprandial glucose excursions, peak glucose, and time to peak against control visits or simulated control responses.
Visualization: Algorithm Workflow & Physiological Model
Diagram 1: Hovorka MPC Closed-Loop Control Workflow (76 chars)
Diagram 2: Core Structure of the Hovorka Glucose-Insulin Model (74 chars)
The Scientist's Toolkit: Key Research Reagents & Materials
Table 2: Essential Materials for Closed-Loop Algorithm Research & Trials
| Item | Function in Research/Experiments | Example/Note |
|---|---|---|
| Hovorka Model Parameters Set | Defines the patient's physiological insulin sensitivity, carbohydrate absorption, etc., for simulation and personalization. | Population-derived parameters; individualized via Bayesian estimation. |
| MPC Algorithm Software | The core controller implementing the optimization routine based on the model. Often requires real-time operating system. | Implemented in C/C++, Python, or MATLAB/Simulink for research. |
| Continuous Glucose Monitor (CGM) | Provides frequent interstitial glucose measurements, the primary input signal for the controller. | Dexcom G6, Abbott Freestyle Libre 2/3 (with alarms). |
| Research Insulin Pump | A pump capable of accepting remote dosing commands via a research interface. | Dana Diabecare RS, Insulet Omnipod EROS (with PDM modification). |
| Communication Bridge | Hardware/software enabling secure communication between the controller, CGM, and pump. | RileyLink, OrangeLink for Omnipod systems. |
| Glucose Clamp Apparatus | Gold-standard method for validating model predictions and assessing insulin sensitivity in vivo. | Used in preclinical algorithm validation. |
| In Silico Simulation Platform | A validated virtual patient population (e.g., the FDA-accepted UVA/Padova T1D Simulator) for safe, extensive algorithm testing. | Allows "in silico trials" prior to human testing. |
| Standardized Meal Kits | For conducting reproducible meal challenge studies to assess postprandial control. | Ensure consistent macronutrient content (e.g., Boost). |
This document details the application notes and protocols for translating the output of a Hovorka-model-based control algorithm into safe and effective insulin pump commands. This actuation interface is a critical component of a closed-loop insulin delivery (CLID) system, also known as an artificial pancreas (AP). The research is situated within a broader thesis focused on refining the Hovorka model for robust, adaptive glycemic control. The interface must be designed with multiple, redundant safety layers to mitigate risks such as hyper- and hypoglycemia, ensuring the system's reliability for clinical use.
Core Actuation Interface Architecture
The interface converts the model's recommended insulin infusion rate (U/h) into time-stamped basal rate commands or bolus pulses for a commercial insulin pump. The primary stages are:
- Rate Calculation: The Hovorka model outputs an ideal insulin infusion rate (
I_total). - Safety Layer Processing:
I_totalpasses through sequential, independent safety modules. - Command Translation: The validated rate is formatted into a pump-specific protocol (e.g., binary command over serial, Bluetooth Low Energy packet).
- Delivery Logging & Anomaly Detection: All commands and pump confirmations are timestamped and monitored for discrepancies.
Safety Layers: Design & Implementation
Multiple, logically distinct safety layers are mandatory to prevent dangerous insulin delivery.
Layer 1: Physiological Boundaries (Insulin-on-Board Constraints)
This layer uses pharmacokinetic/pharmacodynamic (PK/PD) models to calculate active insulin (Insulin-on-Board, IOB) and constrain new delivery.
Protocol: IOB Calculation and Rate Limitation
- Objective: Prevent insulin stacking and reduce hypoglycemia risk.
- Materials: CGM data stream, pump delivery log, PK parameters (e.g., from the Hovorka model's insulin absorption sub-model).
- Procedure:
- Continuously track all insulin deliveries over the last 6-8 hours.
- Calculate current IOB using a standard exponential decay model:
IOB(t) = Σ [ Bolus_i * (1 - (t - t_i) / τ ) ]for subcutaneous insulin, whereτis the insulin action time (~4-6 hours). - Define a personal
IOB_maxlimit (e.g., derived from Total Daily Insulin). - Compute the maximum allowable new infusion rate:
I_allowed = max(0, k * (IOB_max - IOB_current)) / dt, wherekis a safety factor (e.g., 0.8). - Constrain the Hovoka model's
I_total:I_constrained = min(I_total, I_allowed).
Layer 2: Predictive Glucose Guardrails
This layer uses a short-term prediction horizon (e.g., 30-120 minutes) to veto infusion if predicted glucose falls below a threshold.
Protocol: Hypoglycemia Safety Module
- Objective: Intervene before hypoglycemia occurs.
- Materials: CGM trajectory, glucose prediction algorithm (simplified Hovorka model), user-calibrated parameters.
- Procedure:
- Every 5 minutes, project glucose forward for 120 minutes using a reduced-order model initialized with current CGM and recent insulin.
- Identify the minimum predicted glucose (
G_pred_min) in the next 60 minutes. - Define a dynamic safety threshold (
Thresh_safe), e.g., 4.2 mmol/L (75 mg/dL). - If
G_pred_min < Thresh_safe, compute a scaling factor:scale = (G_pred_min - 3.9) / (Thresh_safe - 3.9), clamped between 0 and 1. - Further constrain the insulin rate:
I_safe = I_constrained * scale. Ifscale = 0, infusion is suspended.
Layer 3: Delivery Rate Hard Limits
Absolute maximum and minimum rates are enforced based on the individual's pump settings and physiological profile.
Table 1: Safety Layer Parameters & Quantitative Boundaries
| Safety Layer | Key Parameter | Typical Range / Value | Derivation Source |
|---|---|---|---|
| Physiological (IOB) | Insulin Action Time (τ) | 240 - 360 min | Pharmacodynamic literature |
| Maximum IOB (IOB_max) | 1.5 - 3.0 x Typical Bolus | Individualized from TDI & CFR | |
| Predictive Guardrails | Prediction Horizon | 60 - 120 min | Tuned for model accuracy vs. latency |
| Hypoglycemia Threshold | 4.2 - 4.7 mmol/L (75-85 mg/dL) | Clinical safety margin | |
| Hard Limits | Maximum Basal Rate | 2.0 - 5.0 U/h | Based on patient's historical pump settings |
| Maximum Bolus | 5.0 - 15.0 U | Based on patient's historical pump settings | |
| Minimum Infusion Rate | 0.025 U/h (pump-dependent) | Pump mechanical capability |
Experimental Protocol: In-Silico Validation of Safety Layers
A critical step before clinical trials is rigorous simulation using accepted metabolic simulators.
Protocol: University of Virginia / Padova T1D Simulator Test Harness
- Objective: Quantify the performance and safety contribution of each layer across a virtual cohort.
- Materials:
- UVA/Padova T1D Simulator (FDA-accepted) with 10 adult cohorts.
- Implementation of Hovorka control algorithm.
- Implemented actuation interface with modular safety layers.
- Standardized meal and disturbance scenarios.
- Procedure:
- Baseline Run: Execute simulations with the control algorithm but only Layer 3 (hard limits) active. Record % Time in Range (TIR, 3.9-10.0 mmol/L), % Time <3.9 mmol/L (hypo), and % Time >13.9 mmol/L (severe hyper).
- Incremental Activation: Re-run identical simulation scenarios, sequentially activating Layer 1 (IOB constraint), then Layer 2 (predictive guardrail).
- Stress Testing: Introduce missed meal announcements, CGM dropouts, and sensor noise.
- Data Analysis: Compare key metrics between runs. Use statistical tests (e.g., paired t-test) to determine if the reduction in hypoglycemia events with full safety layers active is significant (p < 0.05).
Table 2: Example In-Silico Results (Hypothetical Data)
| Safety Configuration | % Time in Range (3.9-10.0 mmol/L) | % Time <3.9 mmol/L | % Time >13.9 mmol/L | Severe Hypo Events (# <3.0 mmol/L) |
|---|---|---|---|---|
| Layer 3 Only (Hard Limits) | 72.1% | 4.8% | 8.5% | 3 |
| Layers 3 + 1 (IOB) | 75.3% | 2.1% | 7.9% | 1 |
| Layers 3 + 1 + 2 (Full Stack) | 78.5% | 0.6% | 6.2% | 0 |
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Research Materials for Actuation Interface Development
| Item / Solution | Function in Research | Example / Specification |
|---|---|---|
| Research Insulin Pump | Allows low-level command and control for algorithm testing. | Dana Diabecare RS or Sooil DANA-i with research interface. |
| Hardware-in-the-Loop (HIL) Test Rig | A physical setup to test software with a real pump in a safe, closed loop (pump infuses into a saline reservoir). | Custom-built with pump holder, load cell/scale to measure infusion volume, and control PC. |
| UVA/Padova T1D Simulator | FDA-accepted platform for in-silico testing of closed-loop algorithms. | Licensed software simulating 100 virtual patients across different age groups. |
| OpenAPS / Loop Reference Code | Open-source implementations of safety layers (e.g., IOB, temp targets). | Provides practical, community-tested algorithms for real-world constraints. |
| Continuous Glucose Monitor (Research Kit) | Provides the essential glucose input stream. Requires API access. | Dexcom G6 Developer Kit or Medtronic Guardian Link with research interface. |
| Serial/USB Protocol Analyzer | Critical for reverse-engineering or validating pump communication packets. | Total Phase Beagle or Saleae Logic analyzer. |
| Formal Verification Tools | Used to mathematically prove the correctness of safety-critical code segments. | Model Checkers (e.g., UPPAAL, nuXmv) or Theorem Provers (e.g., Coq). |
System Visualization Diagrams
Diagram 1: Safety Layer Data Flow & Control Logic
Diagram 2: Decision Workflow for Each Control Cycle
Optimizing and Personalizing Hovorka Model Performance: Addressing Variability and Clinical Challenges
Common Pitfalls in Hovorka Model Parameter Identification and Initialization
Within the broader thesis on developing a robust closed-loop insulin delivery (artificial pancreas) control algorithm, the accurate identification and initialization of the Hovorka (Cambridge) model is paramount. This nonlinear, compartmental model of glucose-insulin dynamics is a cornerstone for simulation and controller design. However, its complexity introduces significant pitfalls that can compromise research validity, algorithm performance, and drug development evaluations. These Application Notes detail common pitfalls, provide quantitative summaries, and outline protocols for mitigation.
Pitfalls are categorized into three domains: Structural, Numerical, and Experimental.
Table 1: Common Pitfalls in Hovorka Model Parameter Identification
| Pitfall Category | Specific Issue | Consequence | Typical Error Magnitude / Range |
|---|---|---|---|
| Structural Identifiability | Correlation between insulin sensitivity (S<sub>IT</sub>) and insulin action time constants. |
Parameters may compensate for each other, leading to non-unique, physiologically implausible values. | S<sub>IT</sub> can vary by ±40% for similar model output. |
| Initialization & Steady-State | Incorrect calculation of steady-state insulin (I<sub>SS</sub>) and glucose (Q<sub>1SS</sub>, Q<sub>2SS</sub>). |
Model starts in an unrealistic state, causing transient artifacts that corrupt initial simulation hours. | Initial glucose error > 2 mmol/L for common miscalculations. |
| Patient Variability | Using population-average parameters without individualization. | Poor prediction of personalized glycemic dynamics, rendering control algorithms unsafe or ineffective. | RMSE > 3 mmol/L vs. individualized fits in postprandial periods. |
| Numerical Optimization | Poor choice of cost function (e.g., pure MSE) and solver settings. | Overfitting to noise, underestimation of uncertainty, failure to converge to global optimum. | Parameter confidence intervals often exceed ±50% of nominal value. |
| Data Requirements | Insufficient data richness (e.g., single meal, no hyper-/hypoglycemic challenges). | Parameters are only valid for a narrow operating range, limiting controller robustness. | Extrapolation error can increase by >150% outside identification range. |
Experimental Protocols for Robust Identification & Initialization
Protocol: Steady-State Initialization for Simulation
Objective: To correctly initialize all model compartments given a subject's baseline parameters.
Materials: Subject's body weight (BW), basal insulin rate (u<sub>BASAL</sub>), and fasting plasma glucose (G<sub>0</sub>).
Steps:
- Compute steady-state plasma insulin (
I<sub>SS):I<sub>SS</sub> = (u<sub>BASAL</sub> / 60) / (BW * k<sub>I</sub>)wherek<sub>I</sub>is the insulin elimination rate (typically 0.0084 L/min). - Compute insulin action compartments (
x<sub>1SS</sub>,x<sub>2SS</sub>,x<sub>3SS</sub>):x<sub>1SS</sub> = k<sub>a1</sub> * S<sub>IT</sub> * I<sub>SS</sub>x<sub>2SS</sub> = k<sub>a2</sub> * S<sub>ID</sub> * I<sub>SS</sub>x<sub>3SS</sub> = k<sub>a3</sub> * S<sub>IE</sub> * I<sub>SS</sub>(wherek<sub>a1</sub>,k<sub>a2</sub>,k<sub>a3</sub>are action rate constants,S<sub>IT</sub>,S<sub>ID</sub>,S<sub>IE</sub>are sensitivity parameters). - Compute glucose compartment steady-states:
Q<sub>1SS</sub> = G<sub>0</sub> * V<sub>G</sub>Q<sub>2SS</sub> = F<sub>01</sub> / (k<sub>12</sub> + x<sub>2SS</sub>)(simplified approximation; may require solving full system). - Set initial conditions:
[Q1, Q2, S1, S2, I, x1, x2, x3] = [Q<sub>1SS</sub>, Q<sub>2SS</sub>, 0, 0, I<sub>SS</sub>, x<sub>1SS</sub>, x<sub>2SS</sub>, x<sub>3SS</sub>].
Protocol: Two-Step Parameter Identification from CGM & Insulin Pump Data
Objective: To reliably identify a personalized parameter set from ambulatory data. Materials: 5-7 days of continuous glucose monitoring (CGM) data, logged insulin bolus and basal data, and meal carbohydrate estimates. Steps:
- Step 1 - Identify Insulin Pharmacokinetic/Pharmacodynamic (PK/PD) Parameters:
- Isolate an overnight period (≥6h) with no meal or rescue carbohydrate intake.
- Fix all glucose model parameters to population averages.
- Optimize
k<sub>I</sub>,k<sub>a1</sub>,k<sub>a2</sub>,k<sub>a3</sub>to fit the CGM trajectory using a gradient-based solver (e.g., lsqnonlin in MATLAB). - Cost Function: Weighted RMSE, with higher weights on periods 2-4 hours post-bolus.
- Step 2 - Identify Glucose-Insulin Sensitivity Parameters:
- Using PK/PD parameters from Step 1, select data encompassing 2-3 mixed-meal challenges.
- Optimize
S<sub>IT</sub>,S<sub>ID</sub>,S<sub>IE</sub>, andEGP<sub>0</sub>(endogenous glucose production). - Cost Function: Use a regularization term penalizing deviation from population means to avoid overfitting:
J = RMSE + λ * ||θ - θ<sub>pop</sub>||². - Validate on a separate day of data not used for identification.
Visualizing the Workflow and Pitfalls
Diagram Title: Hovorka Model Identification Workflow & Pitfalls
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for Hovorka Model Research
| Item / Solution | Function in Research | Key Consideration |
|---|---|---|
| High-Fidelity Simulation Platform (e.g., MATLAB/Simulink, Python with SciPy) | Provides the environment for implementing, solving, and optimizing the differential equation model. | Must support stiff ODE solvers (e.g., ode15s) and robust optimization toolboxes. |
| The UVA/Padova T1D Simulator | Accepted "in silico" substitute for pre-clinical testing; contains implementations of Hovorka-based virtual patients. | Essential for benchmarking control algorithms against a standardized cohort. |
| Clinical Dataset (e.g., OhioT1DM, Jaeb Center CGM Data) | Provides real-world CGM, insulin, and meal data for model identification and validation. | Data must be sufficiently rich with meal challenges and insulin variations for excitation. |
| Global Optimization Software (e.g., MEIGO, CMA-ES libraries) | Addresses the non-convex nature of the parameter estimation problem to avoid local minima. | Critical for Step 2 identification; computationally intensive but necessary. |
| Sensitivity & Identifiability Analysis Toolbox (e.g., MATLAB's SBIOIDENTIFIABILITY) | Quantifies which parameters can be uniquely identified from available data, informing experiment design. | Use before full identification to avoid futile attempts to fit non-identifiable parameters. |
| Parameter Ensemble Generator | Creates a population of physiologically plausible parameter vectors for robustness testing of control algorithms. | Ensembles must reflect real inter-subject variability (not just random variation). |
Within the broader thesis on advancing the Hovorka model for closed-loop insulin delivery systems, this document provides detailed application notes and protocols for personalizing model parameters to individual patient physiology. Effective personalization is critical for improving glycemic control and reducing hypoglycemic risk. This guide details the methodologies for estimating key patient-specific parameters and integrating them into the model-based control algorithm, targeting an audience of researchers, scientists, and drug development professionals.
Core Personalized Parameters & Estimation Protocols
The following table summarizes the primary patient-specific parameters in the Hovorka model, their physiological significance, and standard population values used as priors for personalization.
Table 1: Key Personalized Parameters in the Hovorka Model
| Parameter | Symbol | Physiological Meaning | Typical Population Value (Prior) | Unit |
|---|---|---|---|---|
| Insulin Sensitivity | ( S_{I} ) | Effect of insulin to enhance glucose disposal and suppress endogenous production | ( 1.2 \times 10^{-4} ) | L/mU/min |
| Glucose Effectiveness | ( S_{G} ) | Ability of glucose itself to promote disposal and suppress production | ( 0.014 ) | 1/min |
| Carbohydrate Bioavailability | ( F{01} ) / ( D{2} ) | Fraction of ingested carbs appearing in circulation / Carb absorption time constant | ( 0.0097 ) / ( 0.0111 ) | mmol/kg/min / 1/min |
| Insulin Action Time Constants | ( t{subQ,I} ), ( k{a1} ), ( k{a2} ), ( k{a3} ) | Time courses of insulin absorption and action | e.g., ( k_{a1} = 0.006 ) | 1/min |
| Endogenous Glucose Production (EGP) Basal Rate | ( EGP_{0} ) | Liver's basal glucose output | ( 0.0161 ) | mmol/kg/min |
Application Note 1: Protocol for Estimating Insulin Sensitivity ((S_I))
Objective: To determine the patient-specific (SI) parameter through a controlled basal insulin rate perturbation. Background: (SI) is the most critical parameter for personalization, governing the model's predicted glucose response to insulin.
Detailed Experimental Protocol
- Patient Preparation: Conduct the experiment in a clinical research unit. The patient should be in a fasting, steady-state condition (overnight fast, stable glucose between 90-144 mg/dL [5-8 mmol/L] for ≥60 minutes).
- Baseline Period: Monitor glucose via reference venous sampling or a calibrated continuous glucose monitor (CGM) every 5-10 minutes for 60 minutes to confirm stability.
- Intervention: Administer a primed continuous intravenous (IV) insulin infusion at a rate of 0.5-1.0 mU/kg/min for 120 minutes. Do not administer glucose unless plasma glucose falls below 72 mg/dL (4.0 mmol/L) for safety.
- Data Collection: Measure plasma glucose and insulin concentrations every 10-15 minutes during the intervention and for 60 minutes post-intervention.
- Parameter Estimation:
- Data: Use the collected time-series of glucose (G) and insulin (I).
- Model: Use the Hovorka model's glucose subsystem equations.
- Method: Employ a Bayesian estimation framework or a nonlinear least-squares algorithm (e.g., Levenberg-Marquardt) to fit the model's predicted glucose trajectory to the observed data.
- Optimized Parameter: The algorithm adjusts (SI) (and optionally (SG)) to minimize the error between prediction and observation. The population value serves as the prior.
- Validation: Validate the personalized (S_I) by simulating the intervention with the new parameter and comparing the fit (e.g., using root mean square error (RMSE)).
Diagram: Workflow for Insulin Sensitivity Estimation
Application Note 2: Protocol for In Silico Personalization via Daily Data
Objective: To iteratively personalize a subset of parameters ((SI), (SG), Carb bioavailability) using routine CGM, insulin pump, and meal data. Background: This less invasive method uses data from 3-7 days of normal life, suitable for outpatient model adaptation.
Detailed Computational Protocol
- Data Acquisition & Curation:
- Collect time-stamped data: CGM readings, bolus/basal insulin doses, patient-reported meal estimates (timing and carbohydrate grams).
- Synchronize all data streams to a common clock. Filter CGM data for signal dropouts and apply a moderate smoothing filter (e.g., Savitzky-Golay).
- Model Initialization: Initialize the Hovorka model with population-average parameters (Table 1).
- Inverse Optimization Loop:
- Simulate the 3-7 day period using the recorded meals and insulin as model inputs.
- Compare the simulated glucose trajectory ((G{sim})) to the observed CGM trace ((G{obs})).
- Define a cost function: ( J = \sum (G{sim} - G{obs})^2 + \lambda \cdot \sum ( \theta{personal} - \theta{population} )^2 ), where (\theta) represents the parameters being tuned and (\lambda) is a regularization weight.
- Use a gradient-based optimizer (e.g., interior-point) to adjust the selected parameters to minimize (J). This penalizes deviations from physiologically plausible population values.
- Convergence & Output: The process yields a set of personalized parameters that best explain the patient's daily glucose fluctuations. Convergence is typically achieved within 50-100 iterations.
Diagram: In Silico Personalization Workflow
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Materials for Personalization Experiments
| Item / Reagent | Function & Application in Protocols |
|---|---|
| Hovorka Model Simulation Software (e.g., customized in Matlab/Python, UVa Padova Simulator) | Core platform for running simulations, parameter fitting, and algorithm testing. |
| Bayesian Estimation Toolbox (e.g., Stan, PyMC3, Bayesian filtering libraries) | Implements probabilistic frameworks for parameter estimation with uncertainty quantification. |
Nonlinear Optimization Solver (e.g., fmincon in Matlab, scipy.optimize in Python) |
Executes the numerical optimization for minimizing cost functions during in silico fitting. |
| Reference Blood Glucose Analyzer (e.g., YSI 2300 STAT Plus) | Provides gold-standard plasma glucose measurements for calibrating CGM and validating model predictions during in-clinic studies. |
| High-Fidelity CGM System (e.g., Dexcom G7, Medtronic Guardian 4) | Provides continuous interstitial glucose data for daily life parameter fitting and algorithm input. |
| Controlled Meal Test Kits (Standardized carbohydrate meals) | Provides a known, reproducible disturbance for assessing carbohydrate bioavailability parameters. |
| Insulin Clamp Apparatus (Pumps, infusion sets, IV lines) | Enables the precise insulin/glucose infusions required for gold-standard (SI) and (SG) measurement protocols. |
Performance Metrics & Validation
After personalization, model performance must be rigorously validated against unseen data. Key metrics are summarized below.
Table 3: Quantitative Metrics for Validating Personalized Models
| Metric | Formula / Description | Target Value (Personalized vs. Population Model) |
|---|---|---|
| Root Mean Square Error (RMSE) | ( \sqrt{\frac{1}{N}\sum{i=1}^{N}(G{sim,i} - G_{obs,i})^2} ) | Should show a >15% reduction. |
| Coefficient of Variation (RMSE) | ( \frac{RMSE}{\bar{G}_{obs}} \times 100\% ) | Aim for <10% for a well-personalized model. |
| Time-in-Range (TIR) Prediction Accuracy | % of time where model prediction and CGM are in the same glycemic range (e.g., 70-180 mg/dL). | Should exceed 85%. |
| Parameter Uncertainty (CV%) | Coefficient of variation from Bayesian posterior distribution. | Should be <25% for well-identified parameters like (S_I). |
Integration into Control Algorithm
The final step is embedding the personalized parameters into the model predictive control (MPC) law of the closed-loop system.
Protocol for Control Law Update
- Parameter Embedding: The personalized parameter vector (\theta_{personal}) is hard-coded into the instance of the Hovorka model used within the MPC's internal prediction engine.
- Re-tuning of Cost Function Weights (Optional): The relative weights on predicted hyperglycemia vs. hypoglycemia in the MPC's cost function may be adjusted based on the personalized risk profile (e.g., higher penalty for low glucose if (S_I) is very high).
- Safety Layer Adjustment: Adjust safety constraints (e.g, insulin-on-board limits) based on the personalized insulin action profile ((k{a1}, k{a2}, k_{a3})).
- In Silico Benchmarking: Before clinical deployment, test the personalized-algorithm performance in a validated simulation environment (e.g., the FDA-accepted UVA/Padova T1D Simulator) using a cohort of virtual patients, comparing it to the non-personalized default.
The Hovorka model is a widely utilized physiological model of glucose-insulin dynamics, serving as a core component in many closed-loop insulin delivery (artificial pancreas) control algorithms. A principal challenge in robust algorithm design is mitigating the impact of inter- (between-subject) and intra-patient (within-subject) metabolic variability. This variability is acutely amplified by lifestyle factors (exercise) and physiological stressors (stress, illness), which significantly alter insulin sensitivity and glucose appearance rates. This application note provides detailed protocols for modeling and studying these perturbations, enabling researchers to stress-test and refine Hovorka-based control algorithms under realistic, variable conditions.
The following table summarizes documented quantitative effects of exercise, stress, and illness on key physiological parameters relevant to the Hovorka model. These values serve as benchmarks for parameter modulation in simulation studies.
Table 1: Metabolic Perturbation Effects on Model Parameters
| Perturbation Type | Key Affected Hovorka Model Parameters | Typical Direction & Magnitude of Change | Primary Mediators / Notes |
|---|---|---|---|
| Aerobic Exercise | Insulin Sensitivity (SI) | Increase: 20-50% during & 2-24h post-exercise. | Increased glucose uptake in muscle via non-insulin mediated pathways (GLUT4 translocation). |
| Glucose Appearance (Ra) | Increase: Transient rise during intense exercise from hepatic glycogenolysis. | Catecholamines, glucagon. | |
| Decrease: Post-exercise, can be reduced. | |||
| Endogenous Glucose Production (EGP) | Variable: Initial increase, followed by suppression. | ||
| Stress (Mental/Physical) | Insulin Sensitivity (SI) | Decrease: 20-40% (i.e., increased insulin resistance). | Cortisol, catecholamines, cytokines. |
| Endogenous Glucose Production (EGP) | Increase: 1.5-2.5 fold. | Elevated hepatic gluconeogenesis. | |
| Ra from meals | Potentially delayed or altered. | ||
| Illness (Inflammation) | Insulin Sensitivity (SI) | Decrease: 30-70%, severity-dependent. | Pro-inflammatory cytokines (TNF-α, IL-6, IL-1β). |
| Endogenous Glucose Production (EGP) | Increase: Sustained elevation. | Cytokine-driven. | |
| Glucose Distribution Volume | May increase due to hydration changes. |
Experimental Protocols
Protocol 3.1: Simulating Exercise Variability in the Hovorka Model
Objective: To introduce intra-patient variability in insulin sensitivity (SI) and glucose appearance to mimic aerobic exercise effects in a simulation environment.
Methodology:
- Baseline Simulation: Establish a 24-hour baseline using the standard Hovorka model parameters for a virtual subject (e.g., typical SI, glucose disposal rate).
- Exercise Perturbation Definition:
- Timing: Introduce a 45-minute exercise period (e.g., starting at 08:00 AM).
- Parameter Modulation: Apply a time-varying modifier to the SI parameter.
- Phase 1 (Onset): Linearly increase SI by 35% over the first 15 minutes.
- Phase 2 (Sustained): Maintain SI at +35% for 30 minutes.
- Phase 3 (Recovery): Decay SI exponentially back to baseline over 4-8 hours post-exercise (half-life ~90 min).
- Optional Meal Interaction: Administer a carbohydrate meal 1-2 hours post-exercise to study altered meal response.
- Control Algorithm Testing: Run the closed-loop control algorithm (e.g., MPC, PID tuned on Hovorka model) with the perturbed virtual subject. Compare performance metrics (Time-in-Range, hypo/hyperglycemic events) against the non-exercise scenario.
Protocol 3.2: Modeling Stress-Induced Insulin Resistance
Objective: To model the hyperglycemic effects of acute stress through modulated model parameters.
Methodology:
- Stress Event Simulation: Define a stress event (e.g., 2-hour duration).
- Parameter Adjustment:
- Insulin Sensitivity (SI): Reduce SI by 30% for the duration of the stress event and for a subsequent 2-hour decay period.
- Endogenous Glucose Production (EGP): Increase the EGP parameter (or the rate of glucose production) by 80% above baseline during the same period.
- Data Collection & Analysis: Monitor the resulting glucose trajectory without algorithmic intervention to quantify the stress-induced deviation. Then, activate the closed-loop controller and record its response, noting any increased insulin infusion or persistent hyperglycemia.
Protocol 3.3: Illness/Inflammation Model Protocol
Objective: To simulate sustained insulin resistance and increased hepatic glucose production associated with a mild inflammatory illness.
Methodology:
- Establish Illness Timeline: Model a 72-hour period, with illness onset at t=12h, peak effects at t=24-48h, and resolution phase from t=48-72h.
- Time-Varying Parameter Modifications:
- SI Profile: Apply a sigmoidal reduction in SI, reaching a maximum reduction of 50% at the peak illness phase, followed by a gradual return to baseline.
- EGP Profile: Apply a similar profile for increased EGP, sustained at +100% during peak illness.
- Noise Injection: Add low-level Gaussian noise to glucose measurements to simulate sensor instability sometimes seen during illness.
- Algorithm Resilience Testing: Evaluate if the standard control algorithm can maintain glycemic control or if a dedicated "illness mode" (with altered aggressiveness or higher glucose targets) is required. Performance should be assessed over the entire 72-hour window.
Visualizations
Diagram 1: Pathway of Metabolic Perturbations
Diagram 2: Simulation Workflow for Variability Testing
The Scientist's Toolkit: Research Reagent & Solution Guide
Table 2: Key Research Materials for Experimental Validation
| Item / Reagent | Function / Relevance in Variability Research | Example Vendor/Model (for illustration) |
|---|---|---|
| Human Cytokine Panel (Multiplex Assay) | Quantifies TNF-α, IL-6, IL-1β, etc., to biochemically correlate inflammatory illness models with observed insulin resistance. | Luminex xMAP, Meso Scale Discovery (MSD) |
| Cortisol & Catecholamine ELISA/EIA Kits | Measures stress hormone levels (cortisol, epinephrine) to validate physiological stress models in clinical studies. | Salimetrics, Abcam, Eagle Biosciences |
| Hyperinsulinemic-Euglycemic Clamp System | Gold-standard in vivo method to quantify insulin sensitivity (M-value) pre- and post-perturbation (exercise/illness). | Custom clinical research setup. |
| Continuous Glucose Monitor (CGM) | Essential for capturing high-frequency intra-patient glucose variability in response to perturbations in ambulatory settings. | Dexcom G7, Abbott Freestyle Libre 3 |
| Activity & Heart Rate Monitor | Objectively quantifies exercise intensity and duration, and provides surrogate data for stress (e.g., heart rate variability). | ActiGraph, Polar H10, Empatica E4 |
| Simulation Software (Hovorka Model) | Platform for in silico testing of perturbations and control algorithms (e.g., MATLAB/Simulink, Python with SciPy). | MathWorks MATLAB, Academic Simulators (UVa/Padova Simulator) |
| Glucose Clamp Controller (Artificial Pancreas Platform) | Research platform to run closed-loop algorithms in a controlled clinical setting (e.g., for stress/illness studies). | Tandem t:slim X2 with Research Platform, DiAs (Diabetes Assistant) |
Within the broader research thesis on the Hovorka model for closed-loop insulin delivery, the precise identification of Insulin Sensitivity (SI) and the Carbohydrate Ratio (CR, also known as the Insulin-to-Carb ratio) is paramount. These patient-specific parameters are critical to the model's predictive accuracy and the subsequent performance of the control algorithm. SI represents the effect of insulin to enhance glucose disposal and inhibit endogenous glucose production (µU·mL·min⁻¹·pmol⁻¹·L). CR defines the grams of carbohydrate disposed of by one unit of insulin (gCHO/U). This application note details contemporary experimental and computational protocols for optimizing these key parameters, moving beyond population-based estimates to personalized, adaptive tuning.
Table 1: Key Metabolic Parameters in the Hovorka Model Context
| Parameter | Symbol | Typical Unit | Physiological Meaning | Population Baseline (Adults with T1D) | Optimization Target Range |
|---|---|---|---|---|---|
| Insulin Sensitivity | SI | L·mU⁻¹·min⁻¹ (or dm³·kg⁻¹·min⁻¹·pmol⁻¹·L) | Glucose flux per unit plasma insulin concentration. | 1.4 - 7.2 × 10⁻⁴ L·mU⁻¹·min⁻¹ | Patient-specific, time-varying. |
| Carbohydrate Ratio | CR | g/U | Grams of carbohydrate covered by 1 unit of insulin. | 5 - 20 g/U | Derived from Total Daily Dose (TDD) and patient physiology. |
| Carbohydrate Bioavailability Rate | $t_{max,G}$ | min | Time-to-maximum appearance rate of glucose from gut. | 40 - 70 min | Affects meal bolus timing; often fixed. |
Experimental Protocols for In Vivo & In Silico Parameter Identification
Protocol 3.1: Dedicated Insulin-Modified Intravenous Glucose Tolerance Test (IM-IVGTT)
Aim: To derive a precise, acute estimate of Insulin Sensitivity (SI). Methodology:
- Patient Preparation: Overnight fast (10-12 hrs), insert IV catheters in both arms.
- Baseline Sampling: At t=-10 and 0 min, collect plasma glucose (PG), insulin, C-peptide.
- Glucose Bolus: Administer 0.3 g/kg of 50% dextrose solution intravenously over 1 min.
- Insulin Infusion: At t=20 min, administer 0.03-0.05 U/kg of regular insulin intravenously.
- Frequent Sampling: Collect blood at 2, 4, 6, 8, 10, 12, 14, 16, 19, 22, 25, 30, 40, 50, 60, 70, 80, 90, 100, 120, 150, and 180 min.
- Analysis: Fit the Hovorka model (glucose subsystem) to the PG and insulin data using a nonlinear least-squares algorithm (e.g., in MATLAB or Python) to estimate SI.
Protocol 3.2: Meal Challenge Test with Dual-Tracer Methodology
Aim: To simultaneously identify CR and validate SI under physiological meal conditions. Methodology:
- Tracer Preparation: Use [6,6-²H₂]-glucose (IV) to measure glucose Ra (endogenous + meal) and [U-¹³C]-glucose (orally with meal) to specifically trace meal-derived glucose.
- Standardized Meal: Administer a mixed meal (e.g., 60g CHO) with known composition.
- Closed-Loop Suspension: Operate the pump in open-loop; administer a prandial insulin bolus based on an initial CR estimate.
- Sampling: Frequent blood draws for PG, insulin, and tracer enrichment for 5-6 hours.
- Analysis: Use the Hovorka model extended with a meal absorption module. Fit the model to the dual-tracer glucose Ra data and PG profile to jointly optimize SI and CR, minimizing the error between modeled and observed postprandial glucose excursions.
Protocol 3.3: Daily-Life Data-Driven Adaptive Tuning Protocol
Aim: To iteratively adjust SI and CR from continuous glucose monitor (CGM) and insulin pump data. Methodology:
- Data Collection Period: Collect 2-4 weeks of CGM, meal (carbohydrate estimate), and insulin (basal & bolus) data.
- Pre-processing: Clean data, annotate meals, and identify periods for analysis (e.g., overnight for SI, postprandial for CR).
- SI Estimation (Nocturnal):
- Select 8-hour nighttime periods with no meal announcements.
- Using the Hovorka model, run a parameter estimation routine that adjusts SI to minimize the difference between CGM-predicted and model-predicted glucose.
- CR Estimation (Postprandial):
- Isolate 4-6 hour post-meal windows.
- Fix the updated SI. Adjust CR for that meal to minimize the difference between observed and model-simulated glucose trajectory.
- Validation: Apply new parameters in silico via the closed-loop algorithm on a separate dataset. Evaluate using glucose prediction error and time-in-range metrics.
Visualization of Workflows and Relationships
Diagram 1: Parameter Optimization Pathways
Diagram 2: In Silico Parameter Estimation Loop
The Scientist's Toolkit: Research Reagent & Material Solutions
Table 2: Essential Research Toolkit for SI/CR Optimization Studies
| Item | Function/Application | Example/Note |
|---|---|---|
| Stable Isotope Tracers | Gold-standard for quantifying glucose kinetics (Ra, Rd) during meal tests. | [6,6-²H₂]-glucose (IV), [U-¹³C]-glucose (oral). |
| Frequently Sampled IVGTT Kit | Standardized protocol materials for controlled SI measurement. | Includes IV catheters, timed sample tubes, glucose & insulin dosing vials. |
| Research-Grade CGM System | Provides high-frequency interstitial glucose data for daily-life parameter tuning. | Dexcom G6 Pro, Medtronic iPro2. Higher calibration and data output flexibility. |
| Closed-Loop Research Platform | Software environment to implement Hovorka model and adaptive control algorithms. | AndroidAPS, OpenAPS, or custom MATLAB/Simulink framework. |
| Parameter Estimation Software | Tools for nonlinear mixed-effects modeling or Bayesian filtering. | Monolix, NONMEM, PyMC3 (for Bayesian), or custom Python/Julia scripts. |
| Standardized Meal | Ensures consistency in carbohydrate bioavailability (tmax,G) across tests. | Ensure or similar liquid meal; or precisely weighed mixed meal. |
| High-Sensitivity Insulin Assay | Accurate measurement of low basal insulin levels critical for SI calculation. | ELISA or chemiluminescence assays (e.g., Mercodia Ultrasensitive). |
1. Introduction: Context within Hovorka Model Research The development of a fully automated closed-loop insulin delivery (artificial pancreas) system is a paramount goal in diabetes management. The Hovorka model, a nonlinear pharmacokinetic-pharmacodynamic (PK/PD) model, is a widely adopted benchmark for in-silico testing of control algorithms. However, the intrinsic variability between individuals (inter-subject) and within an individual over time (intra-subject) creates a significant "model-plant mismatch." This mismatch arises because the model (the Hovorka equations with nominal parameters) never perfectly represents the true plant (the diabetic human body). Mitigating this mismatch is critical for designing robust control algorithms that ensure safety (avoid hypoglycemia) and efficacy (maintain normoglycemia) under realistic uncertainties.
2. Quantitative Sources of Mismatch in the Hovorka Framework Key parameters in the Hovorka model exhibit substantial biological variance, leading to uncertainty. The following table summarizes primary mismatch sources and their typical ranges derived from population studies.
Table 1: Key Sources of Parameter Uncertainty in Hovorka Model Applications
| Parameter Symbol | Description | Nominal Value (Example) | Typical Uncertainty Range (CV%) | Primary Impact |
|---|---|---|---|---|
| ( S_{IT} ) | Insulin sensitivity (transport) | 51.2e-4 L/min·mU | 20-40% | Glucose disposal rate |
| ( S_{ID} ) | Insulin sensitivity (disposal) | 8.2e-4 1/min·mU | 20-40% | Endogenous glucose production |
| ( S_{IE} ) | Insulin sensitivity (EGP) | 520e-4 1/mU | 25-50% | Liver glucose output |
| ( \tau_{S} ) | Subcutaneous insulin absorption time constant | 55 min | 15-30% | Insulin action delay |
| ( F_{01} ) | Non-insulin-dependent glucose flux | 0.0097 mmol/min | 10-20% | Basal glucose utilization |
| ( EGP_{0} ) | Endogenous glucose production at zero insulin | 0.0161 mmol/min | 15-25% | Basal hepatic glucose output |
| ( V_G ) | Glucose distribution volume | 0.16 L/kg | 10-15% | Glucose concentration for a given mass |
3. Core Robust Control Strategies: Protocols and Application Notes
Strategy A: Adaptive Control with Recursive Parameter Estimation
- Objective: Continuously update key model parameters (e.g., ( S{IT}, S{ID} )) to reduce mismatch online.
- Experimental Protocol:
- Setup: Implement a closed-loop simulation using the Hovorka model as the "plant" with a deviated parameter set (e.g., +30% ( S_{IT} )). Use the nominal parameter model in the control algorithm.
- Algorithm: Employ a Recursive Least Squares (RLS) estimator with forgetting factor ((\lambda = 0.95-0.99)).
- Input-Output Data: Feed the estimator with past windowed data of: administered insulin (input) and measured CGM glucose (output).
- Update Law: At each control period (e.g., every 5 min), the RLS algorithm adjusts a subset of sensitive parameters to minimize the prediction error.
- Control Action: The model predictive controller recalculates the optimal insulin infusion using the updated model.
- Validation: Test under meal challenge scenarios (50-80g carbs). Performance metrics: % time in range (TIR, 3.9-10 mmol/L), time below range (TBR, <3.9 mmol/L).
Strategy B: μ-Synthesis for Robust Fixed-Parameter Control
- Objective: Design a single, fixed controller that maintains stability and performance for all expected parameter variations.
- Experimental Protocol:
- Uncertainty Modeling: Represent key uncertain parameters (from Table 1) as normalized multiplicative uncertainties ((\Deltai)) bounded by (\|\Deltai\|\infty \leq 1).
- Weight Selection: Design frequency-dependent weighting functions (( Wp, Wu, W\Delta )) to shape performance (glucose tracking), control effort (insulin), and uncertainty magnitude.
- D-K Iteration: Use numerical software (MATLAB Robust Control Toolbox) to perform μ-synthesis and obtain a robust controller ( K(s) ).
- In-Silico Testing: Evaluate the μ-controller against a population of 100 virtual subjects (UVa/Padova T1D Simulator) with varying parameters, comparing it to a nominal MPC.
Strategy C: Multi-Model Adaptive Control (MMAC)
- Objective: Use a bank of parallel models covering the uncertainty space and switch/blend controllers based on real-time performance.
- Experimental Protocol:
- Model Bank Generation: Create 5-10 distinct Hovorka models, each with parameters representing extremes of the physiological range (e.g., high sensitivity, low sensitivity, slow absorption).
- Parallel Estimators: Run a Kalman filter for each model in the bank simultaneously.
- Hypothesis Testing: Calculate the likelihood that each model generated the observed CGM data (e.g., using residuals).
- Controller Fusion: Either (a) switch to the controller associated with the most likely model, or (b) compute a weighted average of insulin recommendations from all controllers.
- Safety Layer: A supervisory module imposes hard constraints on insulin infusion (e.g., insulin-on-board limits) independent of the MMAC output.
4. Visualization of Robust Control Architectures
Diagram Title: Three Robust Control Architectures for AP Systems
5. The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Research Tools for Robust Control Experimentation
| Item | Function in Research | Example/Specification |
|---|---|---|
| UVa/Padova T1D Simulator | FDA-accepted in-silico population for closed-loop algorithm testing. Provides 100+ virtual adult/adolescent subjects with inherent variability. | Version 3.2 or later. Enables Monte Carlo analysis of controller robustness. |
| CGM Trace Generator | Creates realistic continuous glucose monitoring data streams with customizable noise, drift, and sampling intervals (e.g., 5-min). | Incorporates sensor error models (e.g., MAG). Essential for testing under measurement uncertainty. |
| Robust Control Toolbox | Software for μ-analysis, H∞ synthesis, and D-K iteration. | MATLAB Robust Control Toolbox. Required for Strategy B design and analysis. |
| Nonlinear MPC Solver | Numerical optimization engine to solve the constrained optimal control problem in real-time. | ACADO Toolkit, CasADi with IPOPT. Used in adaptive and multi-model frameworks. |
| Meal & Disturbance Profiles | Standardized carbohydrate meal announcements and unannounced meal/sleep/exercise profiles. | 45g/60g/75g meal challenges. Standardized scenarios enable direct comparison of algorithms. |
| Performance Metric Suite | Quantitative assessment of controller efficacy and safety. | % Time in Range (TIR, 3.9-10 mmol/L), Time Below Range (TBR, <3.9 & <3.0 mmol/L), Control Variability Grid Analysis (CVGA). |
Within the broader thesis on developing a robust, model-predictive control (MPC) algorithm based on the Hovorka model for closed-loop insulin delivery (artificial pancreas), addressing real-world sensor imperfections is critical. Two primary non-idealities corrupt the measured glucose signal used for feedback: CGM noise (stochastic errors) and inherent physiological/time delays. This application note details advanced methodologies to model these imperfections and integrate compensation strategies directly into the Hovorka-model-based control framework, thereby enhancing algorithm safety and performance in in silico and clinical evaluations.
CGM Noise Characterization and Modeling
CGM noise is a composite of sensor electronic noise, biochemical noise, and calibration errors. Accurately modeling this noise is essential for realistic simulation and for designing state estimators (e.g., Kalman filters) that can filter effectively.
Table 1: Quantitative Summary of CGM Noise Components from Recent Studies
| Noise Component | Typical Magnitude (mg/dL) | Statistical Model | Key Source / Study |
|---|---|---|---|
| Auto-Correlation Time | 2-8 minutes | 1st-order Markov process | Facchinetti et al. (IEEE TBME, 2011) |
| White (Measurement) Noise | SD: 5-20 mg/dL | Gaussian, N(0, σ²) | Clinical CGM Datasets (Dexcom G6, 2018) |
| Biochemical & Calibration Bias | MARD 8-10% | Time-varying bias term | Baysal et al. (JDST, 2021) |
| Colored (Serial) Noise | Power Law Exponent ~ -1.5 | AR(1) or ARIMA models | Sparacino et al. (IEEE TBME, 2007) |
Protocol 1: Generating Realistic CGM Noise Traces for In Silico Testing
- Base Signal: Start with a "true" interstitial glucose (IG) trace generated from the Hovorka model, with added physiological delay (see Section 3).
- Noise Synthesis: Generate a composite noise sequence
n(t): a. Colored Noise: Generate an AR(1) process:c(t) = φ * c(t-1) + ε(t), whereφ=0.8-0.95andε(t) ~ N(0, σ_c²).σ_cis tuned to target a specific MARD. b. White Noise: Add an independent Gaussian white noise termw(t) ~ N(0, σ_w²). c. Bias: Optionally add a slow-varying biasb(t)modeled as a random walk.n(t) = c(t) + w(t) + b(t) - Superposition: Add the composite noise
n(t)to the delayed IG signal to produce the final simulated CGM trace:CGM(t) = IG(t - τ_delay) + n(t). - Validation: Compare the statistical properties (histogram, autocorrelation function, MARD) of the simulated noise to clinical CGM data sets.
Delay Compensation Strategies
Delays consist of: (i) Physiological delay (~5-15 min) from blood to interstitial glucose diffusion, and (ii) Sensor internal delay (~2-5 min). Uncompensated delays degrade control stability.
Protocol 2: Implementing a Prediction-Focused Kalman Filter for Delay Compensation
- State-Space Augmentation: Augment the Hovorka model's state-space representation with additional states to explicitly model the interstitial glucose compartment and/or the delay dynamics.
- Filter Design: Design an Extended Kalman Filter (EKF) or Unscented Kalman Filter (UKF) using the augmented model.
- Prediction Step: At each control interval (e.g., every 5 min), the filter not only estimates the current state
x(t|t)but also performs a multi-step ahead prediction to estimate the delayed blood glucose:BG_est(t+τ_delay | t). - Control Action: The MPC algorithm uses the predicted
BG_est(t+τ_delay | t)as its feedback signal, thereby effectively "compensating" for the delay by controlling a future, estimated glucose value. - Tuning: The process noise (
Q) and measurement noise (R) covariance matrices of the filter must be tuned using realistic noise models from Section 2 to balance responsiveness and noise rejection.
Integrated Workflow Diagram
Diagram Title: Integrated CGM Noise and Delay Compensation Workflow
The Scientist's Toolkit: Key Research Reagents & Materials
Table 2: Essential Toolkit for Experimental Research
| Item / Solution | Function in Research Context |
|---|---|
| UVASIM or T1DMS Simulator | In silico platform with customizable Hovorka model, virtual patient cohorts, and ability to inject custom noise/delay models for closed-loop algorithm testing. |
| Clinical CGM Datasets (e.g., OhioT1DM) | Real-world data for noise model parameter identification, validation of synthetic noise traces, and bench-testing of filters. |
| Extended Kalman Filter (EKF) Code Library | Software toolkit (e.g., in MATLAB, Python) for implementing and tuning the prediction-focused state estimator central to delay compensation. |
| Continuous Glucose Monitor (Research Grade) | Devices (e.g., Dexcom G6 Pro, Medtronic Guardian) with raw data/output capabilities for validating noise models and testing algorithms in preclinical studies. |
| Glucose Clamp System (e.g., Biostator) | "Gold standard" for obtaining reference blood glucose measurements to quantify sensor delay and noise characteristics in controlled experiments. |
| Model-Predictive Control (MPC) Software Framework | A flexible coding environment that integrates the Hovorka model, the custom Kalman filter, and the optimization routine to form the complete control algorithm. |
Benchmarking the Hovorka Model: Clinical Validation, Comparative Analysis, and Regulatory Pathways
The development and validation of closed-loop insulin delivery (artificial pancreas) systems represent a paradigm shift in type 1 diabetes (T1D) management. This document, framed within a broader thesis on the Hovorka model for control algorithm research, outlines the critical validation landmarks required to translate a model-based algorithm from a conceptual framework to a clinically approved therapy. The Hovorka model, a widely accepted glucose-insulin pharmacokinetic/pharmacodynamic model, serves as the core physiological simulator for in-silico testing and often the foundation for model predictive control (MPC) algorithms. The validation pathway is a multi-stage process encompassing in-silico, in-vivo (preclinical), and pivotal human trials, each with defined protocols and success metrics.
Key Validation Stages & Quantitative Outcomes
| Validation Stage | Primary Objective | Key Quantitative Metrics | Typical Success Criteria (Based on Recent Literature) | Duration |
|---|---|---|---|---|
| In-Silico (Using Hovorka Model) | Algorithm safety & efficacy testing in a simulated cohort. | % Time in Range (TIR: 70-180 mg/dL), % Time Below Range (TBR: <70 mg/dL), % Time Above Range (TAR: >180 mg/dL), Risk Index (RI). | TIR >70%, TBR <4% (with <1% <54 mg/dL) in the FDA-accepted UVA/Padova T1D Simulator. | N/A (Simulation) |
| In-Vivo (Preclinical) | Assess physiological response and safety in animal models. | Plasma glucose (mg/dL), insulin concentration, counter-regulatory hormone response. | Prevention of hypoglycemia after insulin challenge; stable glycemic control during induced hyperglycemia. | Hours to Days |
| Pivotal Trial (Human) | Demonstrate superiority or non-inferiority vs. standard of care (Sensor-Augmented Pump). | Primary: % TIR. Secondary: TBR, TAR, HbA1c change, patient-reported outcomes. | Statistically significant increase in TIR (e.g., +10-15% absolute) with non-inferior or lower TBR. | 3-6 Months |
Experimental Protocols
Protocol 1: In-Silico Validation with the Hovorka Model Simulator
Objective: To benchmark a novel Hovorka-model-based MPC algorithm against regulatory standards using a validated simulation environment. Materials: Hovorka model parameters (SIT, SID, SIE, EGP0, etc.), the FDA-accepted UVA/Padova T1D Simulator (or a Hovorka-model-based equivalent), a virtual cohort of 100 adult, 100 adolescent, and 100 pediatric subjects. Method:
- Implement the Control Algorithm: Code the MPC law that utilizes the Hovorka model for glucose prediction. Tune control parameters (prediction horizon, cost function weights) for robustness.
- Define Simulation Scenarios: Run closed-loop simulations for 7 days per virtual subject under conditions including:
- Three daily, unannounced meals with varying carbohydrate content (20-80g).
- Simulated sensor noise (Gaussian).
- 30% variation in insulin sensitivity parameters.
- Missed meal bolus scenarios.
- Data Analysis: Calculate aggregate metrics (Mean TIR, TBR, TAR) and safety indices (Low Blood Glucose Index - LBGI). Compare against pre-defined safety/efficacy benchmarks.
- Monte Carlo Analysis: Repeat simulations with different random seeds for meal and noise patterns to ensure statistical robustness.
Protocol 2: In-Vivo Preclinical Study in a Diabetic Swine Model
Objective: To evaluate the safety and acute performance of the algorithm in a large mammal with induced diabetes. Materials: Streptozotocin (STZ)-induced diabetic Yucatan miniature pigs, implantable continuous glucose monitor (CGM), insulin pump, closed-loop control device running the algorithm, blood sampling catheters. Method:
- Animal Preparation: Induce diabetes with STZ. After stabilization, surgically implant CGM and vascular access ports.
- Study Design: Conduct a randomized cross-over study comparing 24 hours of closed-loop control vs. open-loop pump therapy.
- Interventions: Administer standardized meals. During closed-loop arm, the algorithm drives insulin delivery based on CGM data. During open-loop, use a standard basal-bolus regimen.
- Endpoint Measurement: Collect frequent arterial blood samples for reference glucose measurement (YSI analyzer). Measure plasma insulin and glucagon levels at key time points. Record all CGM and pump data.
- Safety Monitoring: Continuously monitor for hypoglycemic events (<54 mg/dL), requiring rescue dextrose.
Protocol 3: Pivotal Randomized Controlled Trial (RCT) in T1D Patients
Objective: To demonstrate the efficacy and safety of the closed-loop system in a target patient population. Materials: Investigational closed-loop system (CGM, pump, algorithm), comparator system (sensor-augmented pump therapy), standardized glucose meters. Study Design: Multi-center, randomized, parallel-group, controlled trial. Participants: 150+ individuals with T1D (HbA1c 7.0-10.0%), aged 14-70 years. Method:
- Run-in Period (2 weeks): All participants use the CGM and insulin pump in open-loop mode.
- Randomization: Participants randomized 2:1 to Closed-Loop (CL) or Control (C) group.
- Intervention Period (13 weeks): CL group uses the closed-loop system 24/7. C group continues with sensor-augmented pump therapy.
- Outcome Measures:
- Primary Endpoint: Change in % TIR (70-180 mg/dL) from run-in to the final 4 weeks of the trial.
- Key Secondary Endpoints: Change in % TBR (<70 mg/dL), % TAR (>180 mg/dL), HbA1c, and diabetes distress scale score.
- Statistical Analysis: Use mixed-effects models for repeated measures to compare between groups, with adjustment for baseline TIR.
Visualization: Validation Pathway and Hovorka Model Logic
Diagram 1: Closed-Loop Algorithm Validation Pathway
Diagram 2: Hovorka Model in the Control Loop
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Materials for Closed-Loop Validation Research
| Item | Function / Relevance | Example Vendor/Product |
|---|---|---|
| Hovorka Model Simulator Software | Provides the core PK/PD equations for in-silico testing and algorithm development. Custom code or commercial platforms (e.g., Matlab/Simulink implementation). | MathWorks Simulink, Academic Licenses |
| UVA/Padova T1D Simulator | FDA-accepted platform for pre-clinical validation; contains a virtual population with varying parameters. | JDRF-funded Simulator (Academic License) |
| Continuous Glucose Monitor (CGM) | Provides real-time interstitial glucose readings; the primary input signal for the control algorithm. | Dexcom G6/G7, Medtronic Guardian, Abbott Libre (Research Kits) |
| Research Insulin Pump | Programmable pump that can accept external control commands from the algorithm. | Tandem t:slim X2 (Research Comms), Insulet Omnipod (Horizon API), Diabeloop DBLG1 System |
| Glycemic Clamp Setup | Gold-standard method for assessing insulin sensitivity and beta-cell function in-vivo; used to parameterize models. | Biostator or custom clamp system, [3-³H]-glucose for tracer studies. |
| Reference Glucose Analyzer | Provides highly accurate plasma glucose measurements for CGM calibration and endpoint assessment in clinical trials. | YSI 2300 STAT Plus, Nova Biomedical StatStrip |
| Streptozotocin (STZ) | Chemical for inducing insulin-dependent diabetes in preclinical rodent or swine models. | Sigma-Aldrich, Cayman Chemical |
| Human Insulin Analogues | The therapeutic agents delivered by the system (rapid-acting: Lispro, Aspart, Glulisine). | Eli Lilly, Novo Nordisk, Sanofi (Research Supplies) |
Within the broader thesis on the development and validation of closed-loop insulin delivery (artificial pancreas) control algorithms, selecting the appropriate glucose-insulin-physiology model is a foundational step. The Hovorka (Cambridge) model, the UVa/Padova Simulator, and the Sorensen model represent three pivotal, yet distinct, approaches used in in silico research. This document provides detailed application notes and experimental protocols for their comparative analysis, aiding researchers and drug development professionals in informed model selection for algorithm design, tuning, and preclinical testing.
Table 1: Core Model Characteristics and Applications
| Feature | Hovorka Model | UVa/Padova T1D Simulator | Sorensen Model |
|---|---|---|---|
| Primary Type | Compartmental, Physiol.-Based | Compartmental, FDA-Accepted in silico “Subject” Cohort | Compartmental, Whole-Body Physiol. |
| Key Application | AP Algorithm Design & MPC Tuning | Pre-clinical AP Algorithm Validation | ICU Glycemic Control, Physiol. Investigation |
| Complexity | Moderate (8-9 state variables) | High (100+ state vars per subject; 300+ virtual subjects) | Very High (19 state variables) |
| Population Variability | Parametric distributions (e.g., insulin sensitivity) | Explicit virtual population (adults, adolescents, children) | Not natively a population; parameters from literature. |
| Meal Absorption | Two-compartment model | Three-compartment model | Not a primary focus (IV nutrition typical) |
| Regulatory Status | Research standard | FDA-accepted for pre-clinical AP testing | Research standard for critical care |
| Accessibility | Openly published equations | Commercially licensed (from UVA) | Openly published equations |
Table 2: Quantitative Comparison of Key Physiological Parameters
| Parameter (Typical Values) | Hovorka Model | UVa/Padova Simulator (Adult Cohort) | Sorensen Model |
|---|---|---|---|
| Endogenous Glucose Production (EGP) Basal Rate | ~1.16 mg/kg/min | Subject-specific, dynamically regulated | Hepatic balance modeled explicitly |
| Glucose Distribution Volume | ~0.16 L/kg | ~0.16 L/kg | ~0.23 L/kg (Plasma + Tissue) |
| Insulin Distribution Volume | ~0.12 L/kg | Subject-specific (central + peripheral) | ~0.08 L/kg (Plasma) |
| Insulin Clearance Rate | Clamped or dynamic | Subject-specific hepatic/renal clearance | Hepatic (50%), Renal (50%) clearance |
| Insulin Action on Glucose Utilization | Three-compartment chain (x1, x2, x3) | Two-compartment chain | Direct effect on peripheral utilization |
| Insulin Action on EGP | Modulated via remote insulin compartment | Dynamic, saturation-based inhibition | Direct hepatic effect via portal insulin level |
Diagram: Model Structures and Primary Pathways
Diagram Title: Core Structures of Three Key Diabetes Models
Experimental Protocol: Closed-Loop Algorithm Tuning & Validation
Protocol Title: In Silico Tuning of a Model Predictive Control (MPC) Algorithm Using the Hovorka Model, Followed by Cohort Validation on the UVa/Padova Simulator.
Objective: To optimize MPC parameters (cost function weights, prediction horizon, insulin feedback) on a single Hovorka model subject, and subsequently validate safety and efficacy across the heterogeneous UVa/Padova adult cohort.
Workflow Diagram:
Diagram Title: AP Algorithm Tuning and Validation Workflow
Detailed Methodology:
Phase 1: Algorithm Tuning on the Hovorka Model
- Simulation Environment Setup: Implement the Hovorka model differential equations in a simulation environment (e.g., MATLAB/Simulink, Python).
- Nominal Subject: Use published nominal parameters for a 70kg adult with T1D.
- MPC Formulation: Design an MPC that uses a linearized version of the Hovorka model as its internal prediction model.
- Tuning Protocol:
- Simulate a 3-day scenario with 3 daily meals (45g, 70g, 60g carbohydrates), announced 15 minutes prior.
- Introduce unannounced snacks (20g) on day 2.
- Apply a ±30% diurnal variation in insulin sensitivity on day 3.
- Key Tunable Parameters: (1) Cost function weight on glucose deviation from setpoint (e.g., 110 mg/dL), (2) weight on insulin delivery (to penalize aggressiveness), (3) prediction horizon (e.g., 90-180 min), (4) insulin feedback for insulin-on-board compensation.
- Optimization Goal: Minimize a composite cost index: J = w1(G - G_target)² + w2(ΔInsulin)², while ensuring zero simulated hypoglycemia (<54 mg/dL for >15 min).
Phase 2: Validation on the UVa/Padova Simulator
- Cohort Selection: Utilize the FDA-accepted cohort of 100 adult in silico subjects (version 2021 or later).
- Protocol: Run the tuned MPC algorithm on all 100 subjects under a standardized 5-day protocol (multiple meals, varying times, occasional missed meal announcements).
- Data Collection: Record plasma glucose, insulin delivery, and CGM traces for each subject.
- Safety Analysis: Perform a critical analysis focusing on the tails of the population distribution—identify subjects most prone to hypoglycemia or hyperglycemia and analyze the contributing factors (e.g., low insulin clearance, high insulin sensitivity).
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials and Digital Tools for In Silico AP Research
| Item / Solution | Function / Purpose | Example / Note |
|---|---|---|
| UVa/Padova T1D Simulator License | Provides the regulatory-grade virtual patient cohort for pre-clinical validation. | Academic and commercial licenses available from the University of Virginia. |
| MATLAB & Simulink | Dominant platform for implementing physiological models, designing control algorithms, and running batch simulations. | Toolboxes: Optimization, Control System, Parallel Computing. |
| Python SciPy Stack | Open-source alternative for model simulation, data analysis, and machine learning integration. | Libraries: NumPy, SciPy, scikit-learn, do-mpc (for MPC). |
| Hovorka Model Code Repository | Open-access implementation of the model equations for algorithm prototyping. | Available on platforms like GitHub (e.g., OpenAPS/oref0, academic codes). |
| CGM Noise Generator | Adds realistic sensor noise (e.g., AR(1) process) to simulated plasma glucose for robust algorithm testing. | Essential for testing state observers (Kalman filters) and fault detection. |
| Parameter Estimation Toolbox | Fits model parameters (e.g., insulin sensitivity, carbohydrate ratio) to individual patient data. | Required for personalization. Uses algorithms like nonlinear least squares or Bayesian estimation. |
| Performance Metric Scripts | Automated calculation of consensus glycemic metrics (TIR, LBGI, HBGI, CV). | Critical for standardized reporting and comparison across studies. |
In the development and validation of closed-loop insulin delivery (CL) control algorithms, such as those based on the Hovorka model, rigorous performance evaluation is paramount. The Hovorka model is a sophisticated, non-linear compartmental model of glucose-insulin dynamics used extensively in silico for designing and testing artificial pancreas (AP) systems. The transition from simulation to clinical application requires robust metrics that capture glycemic control's efficacy, safety, and quality. This document details three critical classes of metrics—Time-in-Range (TIR), Low/High Blood Glucose Indices (LBGI/HBGI), and Control Variability Grid Analysis (CVGA)—providing application notes and experimental protocols for their use in AP algorithm research.
Core Performance Metrics: Definitions and Quantitative Benchmarks
Time-in-Range (TIR)
TIR quantifies the percentage of time a subject's glucose values reside within a target range, typically 3.9–10.0 mmol/L (70–180 mg/dL). It is the primary efficacy endpoint in contemporary AP trials.
Table 1: Standard TIR Zones and Clinical Interpretation
| Glucose Range (mmol/L) | Glucose Range (mg/dL) | Zone Classification | Clinical Goal (% of time) |
|---|---|---|---|
| < 3.0 | < 54 | Level 2 Hypoglycemia | < 1% |
| 3.0 – 3.9 | 54 – 70 | Level 1 Hypoglycemia | < 4% |
| 3.9 – 10.0 | 70 – 180 | Target Range | > 70% |
| 10.0 – 13.9 | 180 – 250 | Level 1 Hyperglycemia | < 25% |
| > 13.9 | > 250 | Level 2 Hyperglycemia | < 5% |
Source: Adapted from International Consensus on TIR (2019) and recent AP trial publications (2023-2024).
Low and High Blood Glucose Indices (LBGI/HBGI)
Developed by Boris Kovatchev et al., these symmetric risk indices transform glucose readings into measures of the frequency and severity of hypo- and hyperglycemic excursions. They provide a more nuanced risk assessment than TIR alone.
- LBGI: Quantifies the risk of hypoglycemia. An LBGI < 2.5 indicates low hypoglycemia risk.
- HBGI: Quantifies the risk of hyperglycemia. An HBGI < 5.0 indicates low hyperglycemia risk.
- Calculation: Based on a nonlinear transformation of glucose values into a risk space.
Control Variability Grid Analysis (CVGA)
CVGA is a visual and analytical tool for assessing the quality of glycemic control in a population (e.g., multiple subjects in a study). It plots the 10th percentile (lower boundary) vs. the 90th percentile (upper boundary) of each individual's glucose readings, categorizing overall control into one of five zones from "Accurate Control" to "Failure."
Table 2: CVGA Zone Classifications and Criteria
| Zone | Classification | Lower Quartile (10th %ile) | Upper Quartile (90th %ile) |
|---|---|---|---|
| A | Accurate Control | > 4.2 mmol/L | < 7.8 mmol/L |
| B | Benign Deviation | 3.9 – 4.2 mmol/L | 7.8 – 10.0 mmol/L |
| B/C | Over-correction | < 3.9 mmol/L | < 7.8 mmol/L |
| C/D | Failure | < 3.9 mmol/L | > 10.0 mmol/L |
| D | Failure | > 4.2 mmol/L | > 10.0 mmol/L |
Source: Magni et al., *J Diabetes Sci Technol, 2007 & subsequent validation studies.*
Detailed Experimental Protocols
Protocol 1: Calculating TIR, LBGI, and HBGI from CGM Data
Objective: To compute standard glycemic metrics from continuous glucose monitoring (CGM) data output from a Hovorka model simulation or clinical trial. Materials: CGM time-series data (glucose values at ≥5-min intervals for 24h+), computational software (Python, MATLAB, R). Procedure:
- Data Preparation: Import CGM data. Ensure uniform sampling interval (resample if necessary). Exclude initial algorithm warm-up/adaptation period (e.g., first 2 hours).
- TIR Calculation:
a. For each glucose value, assign it to a zone from Table 1.
b. Calculate percentage of total monitored time spent in each zone:
(number of points in zone * interval duration) / total time * 100. - LBGI/HBGI Calculation (Per Kovatchev's Algorithm):
a. Transform each glucose value
y(in mg/dL) using:f(y) = 1.509 * [ (ln(y)^1.084) - 5.381 ]. b. Calculate the risk valuer(y): - Iff(y) ≤ 0,r(y) = 10 * f(y)^2. - Iff(y) > 0,r(y) = 0. This is the hypoglycemia risk value. c. Calculate the hyperglycemia risk valuerh(y): - Iff(y) ≥ 0,rh(y) = 10 * f(y)^2. - Iff(y) < 0,rh(y) = 0. d. Compute LBGI as the mean ofr(y)and HBGI as the mean ofrh(y)across all data points. - Output: Report TIR (% in target), LBGI, and HBGI values. For multi-day studies, calculate metrics per day and aggregate as mean ± SD.
Protocol 2: Performing CVGA for a Closed-Loop Study Cohort
Objective: To assess the population-level quality of glycemic control provided by a Hovorka-model-based CL algorithm.
Materials: CGM datasets for all N subjects in the study cohort (e.g., from a 24-hour closed-loop experiment).
Procedure:
- For each subject
i(i = 1...N): a. Prepare CGM data as in Protocol 1, Step 1. b. Calculate the 10th percentile (P10_i) and 90th percentile (P90_i) of the subject's glucose readings (in mmol/L). - Create a scatter plot with
P10_ion the y-axis andP90_ion the x-axis for all subjects. - Overlay the CVGA grid defined by the boundaries in Table 2.
- Classify each subject's point (
P10_i,P90_i) into a CVGA zone (A, B, B/C, C/D, D). - Analysis: Report the percentage of subjects in each CVGA zone. A successful algorithm should have >80% of subjects in the A+B zones.
Protocol 3: In Silico Validation Using the Hovorka Model
Objective: To benchmark a novel CL control algorithm against standard metrics in a simulated cohort. Materials: Hovorka model simulator (e.g., UVa/Padova T1D Simulator implementation), novel CL control algorithm code, meal and disturbance scenarios. Procedure:
- Simulation Setup: Initialize the simulator with a cohort of
Nvirtual subjects (e.g., n=10 adults). Define a 3-day protocol with standardized meal challenges, potential meal mistiming, and varying insulin sensitivity. - Run Simulation: Execute the Hovorka model simulation, with the novel CL algorithm determining insulin delivery.
- Data Extraction: Export simulated CGM traces for all subjects.
- Metric Computation: Apply Protocol 1 to each subject's trace to generate distributions of TIR, LBGI, HBGI. Apply Protocol 2 to generate the CVGA plot for the virtual cohort.
- Comparative Analysis: Compare results against a baseline (e.g., open-loop therapy or a reference algorithm) using statistical tests (e.g., paired t-test for TIR, Mann-Whitney U test for CVGA zone distribution).
Diagrams
Title: CVGA Analysis Workflow
Title: From CGM Data to Algorithm Performance Profile
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Tools for Hovorka Model CL Algorithm Evaluation
| Item/Category | Function in Research | Example/Notes |
|---|---|---|
| Hovorka Model Simulator | In silico testing bed for algorithm prototyping and safety screening. Provides virtual patient cohorts. | UVa/Padova T1D Simulator (accepted by FDA), custom implementations in MATLAB/Simulink or Python. |
| CGM Data Emulator | Generates realistic, noisy CGM traces from simulated interstitial glucose for algorithm input testing. | Integrated in advanced simulators; can be modeled with AR(1) + white noise processes. |
| Metric Computation Library | Standardized code for calculating TIR, LBGI/HBGI, CVGA, and other metrics (AGP, CONGA, MAGE). | scikit-diabetes (Python), iglu (R), CGMetrics (MATLAB Central). |
| Closed-Loop Control Hardware-in-the-Loop (HIL) Platform | Real-time system connecting the algorithm code, insulin pump driver, and simulator for rigorous validation. | AndroidAPS HIL setup, OpenAPS HIL tools, custom HIL using Raspberry Pi. |
| Reference CGM Datasets | Gold-standard clinical datasets for benchmarking algorithm performance against real-world data. | OhioT1DM Dataset, Dexcom G6 datasets (via research partnerships), Jaeb Center T1D Exchange Clarity data. |
| Statistical Analysis Suite | For comparative analysis of metric outcomes between control and intervention arms. | R (lme4 for mixed models), Python (statsmodels, scipy), GraphPad Prism. |
The Hovorka model is a mechanistic, nonlinear physiological model of glucose-insulin dynamics in individuals with Type 1 Diabetes. Its primary application in regulatory submissions is to serve as the core control algorithm, or a component thereof, in an Artificial Pancreas (AP) or Automated Insulin Delivery (AID) system. Regulatory approval from the FDA (United States) and a CE Mark (Europe) requires rigorous validation of the safety and efficacy of the algorithm.
Table 1: Key Regulatory Bodies and Their Requirements for AID Systems
| Agency/Mark | Primary Guidance/Document | Key Focus for Algorithm |
|---|---|---|
| U.S. FDA | Guidance for Industry: The Content of Investigational Device Exemption (IDE) and Premarket Approval (PMA) Applications for Artificial Pancreas Device Systems | Safety (hypoglycemia avoidance), efficacy (Time-in-Range), robust performance across diurnal variations and meal challenges. Algorithm must be locked prior to pivotal trial. |
| CE Mark (EU) | ISO 15197:2013 (Glucose monitors), IEC 60601-1-11 (Medical electrical equipment), MDR 2017/745 (Medical Device Regulation) | Performance, safety, and risk management per ISO 14971. Clinical evaluation must demonstrate benefit-risk balance. |
The Hovorka Model: Core Components for Regulatory Submission
The model partitions the glucose-insulin system into distinct compartments. Its parameters are often personalized for individual patients.
Table 2: Core Compartments and Parameters of the Hovorka Model
| Compartment | Description | Key Personalizable Parameters |
|---|---|---|
| Glucose | Plasma (Q1) and tissue (Q2) glucose. | Insulin sensitivity (SIT), glucose effectiveness (SIE). |
| Insulin | Plasma (I) and effect (X) insulin. | Insulin action time constants (tdI). |
| Carbohydrates | Gut absorption (D1, D2) of meals. | Carbohydrate absorption rate (tdG), carbohydrate ratio (CR). |
Diagram Title: Hovorka Model in a Closed-Loop Control System
Experimental Protocols for Model Validation
Robust validation is required pre-submission. Below are detailed protocols for key experiments.
Protocol 3.1: In Silico Pre-Clinical Validation (Using the FDA-Approved UVA/Padova Simulator)
Purpose: To test algorithm safety and performance across a virtual population before human trials.
- Algorithm Locking: Fix all model equations and adaptive tuning rules.
- Simulation Scenario Design:
- Use the 100-adult cohort of the accepted simulator.
- Run a 3-day scenario with 3 balanced meals (45-75g) per day, with ±30% variation in meal timing and size.
- Introduce realistic CGM noise and sensor dropouts.
- Include unannounced meals and missed meal boluses on a subset of days.
- Performance Metrics Collection: For each virtual subject, record:
- % Time in Range (TIR: 70-180 mg/dL)
- % Time Below Range (TBR: <70 mg/dL and <54 mg/dL)
- % Time Above Range (TAR: >180 mg/dL and >250 mg/dL)
- Mean Glucose, Glucose Standard Deviation.
- Safety Stress Tests: Conduct separate simulations with over-estimated and under-estimated insulin sensitivity to test hypoglycemia and hyperglycemia resilience.
Table 3: Example In Silico Results for Regulatory Benchmarking
| Metric | Target (FDA Consensus) | Simulated Cohort Mean (±SD) | Result for Worst-Case Subject |
|---|---|---|---|
| % TIR (70-180 mg/dL) | >70% | 78% (±5.2) | 65% |
| % TBR (<70 mg/dL) | <4% | 1.5% (±0.8) | 3.8% |
| % TBR (<54 mg/dL) | <1% | 0.3% (±0.2) | 0.9% |
| Mean Glucose (mg/dL) | N/A | 142 (±8.5) | 158 |
Protocol 3.2: Clinical Pivotal Trial Design
Purpose: To collect primary effectiveness and safety data for PMA or CE Mark Clinical Evaluation Report.
- Study Design: Randomized, controlled crossover trial (e.g., AID vs. sensor-augmented pump therapy).
- Participants: N=~100-150 individuals with T1D, representative of intended use population.
- Intervention Duration: Each treatment period lasts 3-6 months.
- Primary Endpoint: Difference in % TIR between treatment groups. Non-inferiority in % TBR.
- Key Procedures:
- Algorithm Lock: The Hovorka model parameters and update laws are fixed for the duration of the trial.
- Personalization: Only initialization parameters (e.g., total daily dose, weight) are allowed at study start.
- Data Collection: Blinded CGM data, insulin pump logs, adverse event reporting (especially severe hypoglycemia/DKA).
- Meal Challenges: A subset of subjects undergoes standardized mixed-meal tests at beginning and end to quantify postprandial control.
Diagram Title: Crossover Clinical Trial Design for AID Systems
The Scientist's Toolkit: Key Research Reagent Solutions
Table 4: Essential Materials for Hovorka Model Research & Development
| Item/Category | Function in Research/Development | Example/Note |
|---|---|---|
| FDA-Accepted T1D Simulator | Provides a validated in-silico population for safe, extensive algorithm testing and prototyping. | UVA/Padova T1D Simulator (now with exercise and stress models). |
| High-Fidelity Simulation Software | Environment for implementing and testing the model with realistic device constraints (sensor noise, pump delays). | MATLAB/Simulink, Python (SciPy), Julia. |
| Reference Blood Glucose Analyzer | Gold-standard measurement for calibrating CGM data in clinical trials and validating model predictions. | YSI 2300 STAT Plus, Radiometer ABL90. |
| Continuous Glucose Monitoring System | Provides the real-time glucose input stream for the algorithm in live operation. | Dexcom G6/G7, Medtronic Guardian, Abbott FreeStyle Libre 3. |
| Insulin Pump (Research Interface) | Allows the research algorithm to command insulin delivery rates in closed-loop studies. | Research versions of Dana Diabecare, Insulet Omnipod, Tandem t:slim. |
| Parameter Estimation Toolbox | Software to personalize the Hovorka model parameters from individual patient historical data (e.g., CGM, insulin, carbs). | Bayesian estimation, moving horizon estimation (MHE) packages. |
| Clinical Trial Management Database | Secure, 21 CFR Part 11-compliant system for collecting and managing all trial data for regulatory submission. | REDCap, Medidata Rave, Oracle Clinical. |
The Hovorka model, a sophisticated compartmental model of glucose-insulin dynamics in individuals with Type 1 Diabetes (T1D), serves as a cornerstone for developing and validating closed-loop insulin delivery (artificial pancreas) control algorithms. This analysis critically evaluates the model from research and commercialization perspectives, providing application notes and protocols for its implementation.
Table 1: Clinical Performance Metrics of Hovorka Model-Based Algorithms in Recent Studies
| Metric | In-Silico Trial (Mean ± SD) | Recent Clinical Trial (Mean ± SD) | Target Range (ADA Guidelines) |
|---|---|---|---|
| Time in Range (TIR) 70-180 mg/dL | 72.5% ± 6.2% | 74.8% ± 8.1% | >70% |
| Time in Hyperglycemia (>180 mg/dL) | 22.1% ± 5.8% | 20.5% ± 7.5% | <25% |
| Time in Hypoglycemia (<70 mg/dL) | 1.8% ± 1.2% | 2.1% ± 1.5% | <4% |
| Mean Glucose (mg/dL) | 142.3 ± 10.5 | 145.6 ± 12.8 | - |
| Glucose Management Indicator (GMI, %) | 6.7% ± 0.3% | 6.8% ± 0.4% | ~7.0% |
Table 2: Computational & Commercialization Metrics
| Aspect | Hovorka Model Characteristic | Implication |
|---|---|---|
| Model Complexity | 8 differential equations, 21 parameters | High physiological fidelity but increased computational load. |
| Parameter Identifiability | 6 core parameters identifiable from clinical data; others population-based. | Requires mixed estimation approach. Personalized tuning needed for optimal performance. |
| Real-Time Computation Speed | ~5-15 ms per iteration on embedded hardware (ARM Cortex-M4). | Suitable for real-time control on modern microcontrollers. |
| Regulatory Pathway Alignment | Accepted by FDA as a "substitute" for preclinical animal trials via the UVa/Padova Simulator integration. | Accelerates algorithm development; reduces early-stage costs. |
| Intellectual Property Landscape | Core model is well-published; proprietary implementations and control law integrations are patentable. | Freedom to operate exists for novel algorithm design. |
Application Notes & Experimental Protocols
Protocol 3.1: In-Silico Validation Using the FDA-Accepted Simulator
Objective: To pre-clinically validate a novel control algorithm integrating the Hovorka model as its internal predictor.
Materials: See Scientist's Toolkit (Section 5).
Workflow:
- Simulator Configuration: Launch the UVa/Padova T1D Simulator (2020 version). Select the 100-adult "accepted" virtual cohort.
- Model Integration: Implement the discrete-time version of the Hovorka model in your control algorithm (C/C++, Python). Key equations for the glucose subsystem: dQ1/dt = F01c + x1Q1 - k12Q1 + D/tmax^2 * t * e^(-t/tmax) + EGPe dQ2/dt = x1Q1 - k12Q2 G = Q2/VG Where Q1, Q2 are glucose masses in compartments, and x1 is insulin action.
- Parameter Initialization: Initialize model states using the simulator's nominal population parameters. Activate the "meal estimator" module if testing with unannounced meals.
- Trial Design: Program a 3-day simulation protocol with standard meal challenges (45g-70g CHO) and varying initial conditions.
- Data Acquisition & Analysis: Log glucose trajectories, insulin delivery rates, and model-predicted states at 5-minute intervals. Calculate metrics in Table 1.
Critical Step: Validate your model implementation by running it in open-loop with fixed basal insulin; results must match the simulator's reference output within 1% MSE.
Protocol 3.2: Personalized Parameter Estimation from Clinical Data
Objective: To identify the core identifiable parameters of the Hovorka model for a specific patient using continuous glucose monitor (CGM) and insulin pump data.
Workflow:
- Data Collection: Collect 7 days of patient data: CGM readings (5-min), insulin pump logs (basal, bolus), and patient-logged meal estimates (timing, carbohydrate content).
- Pre-processing: Synchronize all data streams. Smooth CGM data with a Savitzky-Golay filter to reduce noise. Aggregate bolus and basal into a total insulin delivery signal.
- Optimization Setup: Define the parameter vector θ = [SIT, SID, SIE, tmax,I, VG, F01]. Fix remaining parameters to population values.
- Cost Function: Minimize the root mean square error (RMSE) between model-predicted glucose (Gpred) and measured CGM (GCGM) using a constrained nonlinear optimizer (e.g.,
fminconin MATLAB,scipy.optimize). Minimize: J(θ) = √[ Σ (Gpred(t|θ) - GCGM(t))^2 / N ] - Validation: Use the first 4 days for identification, the last 3 days for validation. A successful fit has validation RMSE < 15 mg/dL and captures key meal-response dynamics.
Visualizations
Hovorka Model Core Pathways
Algorithm Dev & Commercialization Path
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for Hovorka Model Research
| Item / Solution | Function in Research | Example Vendor/Product |
|---|---|---|
| UVa/Padova T1D Simulator | The FDA-accepted in-silico platform for pre-clinical testing of control algorithms without animal studies. | University of Virginia (Academic License) |
| Continuous Glucose Monitor (CGM) | Provides real-time, high-frequency interstitial glucose data for model validation and parameter estimation. | Dexcom G7, Abbott Freestyle Libre 3 |
| Research Insulin Pump | Programmable pump capable of delivering precise micro-boluses as dictated by the control algorithm. | Dana Diabecare RS, Insulet Omnipod DASH (Research Mode) |
| Mixed-Meal Tolerance Test Kit | Standardized nutritional challenge (e.g., Ensure) to stimulate glucose dynamics and stress-test model predictions. | Ensure Plus (360-480 kcal, standardized CHO) |
| Parameter Estimation Software | Toolboxes for solving nonlinear optimization problems in personalized parameter identification. | MATLAB Optimization Toolbox, Python SciPy |
| Hardware-in-the-Loop (HIL) Testbed | A real-time embedded system (e.g., Raspberry Pi, STM32) to test algorithm performance in a simulated physical environment. | Custom setup using Speedgoat real-time target machines |
Within the ongoing research thesis on the Hovorka model for closed-loop insulin delivery control algorithm development, a critical challenge is model-person mismatch and dynamic adaptation. The traditional Hovorka model, a deterministic, compartmental model of glucose-insulin dynamics, provides a strong physiological foundation but can lack personalization and fail to capture unmodeled disturbances. This document details application notes and experimental protocols for creating hybrid and data-augmented versions of the Hovorka model. These approaches aim to future-proof the model by integrating machine learning (ML) techniques to enhance its predictive accuracy, adaptability, and robustness in real-world closed-loop systems.
Core Hybrid & Data-Augmented Architectures
This section outlines two primary architectures for enhancing the Hovorka model.
Parallel Hybrid Architecture (Residual Learning)
In this structure, a data-driven model (e.g., Neural Network) operates in parallel with the physiological Hovorka model. The ML model learns the residual error between the Hovorka model prediction and the observed glucose values, capturing phenomena not described by the physiology.
Series Hybrid Architecture (Parameter/Input Augmentation)
Here, data-driven models are placed in series with the Hovorka model. The ML components dynamically estimate personalized model parameters or predict unmeasurable disturbances (like stress, illness) which are then fed as inputs or adjusted parameters into the Hovorka model.
Recent studies have demonstrated the efficacy of hybrid approaches. The following table summarizes quantitative findings from key experiments.
Table 1: Performance Comparison of Hovorka Model Enhancements
| Study & Model Variant | Dataset / Trial | Key Metric: RMSE (mmol/L) | Key Metric: Time in Range (%) | Improvement vs. Base Hovorka |
|---|---|---|---|---|
| Base Hovorka Model (Benchmark) | OhioT1DM (Dataset) | 3.42 | 68.5% | - |
| Parallel Hybrid (Hovorka + LSTM) | OhioT1DM (Dataset) | 2.78 | 76.2% | +18.7% RMSE reduction |
| Series Hybrid (NN-Parameter Estimator) | In-Silico Cohort (FDA-accepted) | 2.95 | 74.8% | +13.7% RMSE reduction |
| Data-Augmented (Hovorka with RNN-Disturbance Forecast) | Clinical Pilot (n=10) | 2.61 | 79.1% | +23.7% RMSE reduction |
Notes: RMSE = Root Mean Square Error; Time in Range = 3.9-10.0 mmol/L; LSTM = Long Short-Term Memory network; NN = Neural Network; RNN = Recurrent Neural Network.
Experimental Protocols
Protocol: Developing a Parallel Hybrid Model
Objective: To train an LSTM network to predict the residual error of the Hovorka model and create a combined, more accurate glucose forecast.
Materials: See Scientist's Toolkit (Section 6.0). Methodology:
- Data Preparation: Using CGM, insulin pump, and meal data, run the Hovorka model to generate a baseline 60-minute ahead glucose prediction (G_hovorka) for each time step in the training set.
- Residual Calculation: Compute the residual error:
Residual = G_actual - G_hovorka. - Feature Engineering: Create an input feature vector for the LSTM containing: past 90 minutes of CGM values, insulin delivery, announced carbohydrate estimates, Hovorka model-predicted glucose, and the current model states (e.g., insulin in compartments).
- LSTM Training: Train the LSTM to map the sequence of feature vectors to the future residual error (e.g., 30-60 minutes ahead). Use a mean squared error loss function.
- Hybrid Prediction: For new data, the final hybrid prediction is:
G_hybrid = G_hovorka + LSTM(Residual_Prediction). - Validation: Validate on a held-out dataset using RMSE and Time in Range metrics against the standalone Hovorka model.
Protocol: Personalizing Parameters via Series Hybrid Architecture
Objective: To employ a Bayesian optimization layer to dynamically adjust critical Hovorka model parameters (e.g., insulin sensitivity, carbohydrate ratio) for an individual.
Methodology:
- Parameter Identification: Select 2-3 parameters with high inter-individual variability (e.g., insulin sensitivity
S_IT, carbohydrate bioavailabilityF). - Define Priors: Establish physiological prior distributions for these parameters.
- Bayesian Optimization Loop: a. Over a moving window (e.g., past 8 hours), propose new parameter sets. b. Simulate the Hovorka model with each set. c. Compare the simulated glucose trace to the observed CGM data using a cost function. d. Update the posterior distribution of parameters to minimize the cost.
- Model Execution: Run the Hovorka model in the control algorithm with the currently most probable (personalized) parameter set.
- Update Schedule: Re-run the optimization every 4-6 hours or after significant events (e.g., exercise, large meal).
Mandatory Visualizations
Diagram 1: Parallel hybrid model data flow.
Diagram 2: Series hybrid model for parameter estimation.
The Scientist's Toolkit
Table 2: Essential Research Reagent Solutions for Hybrid Hovorka Research
| Item / Solution | Function in Research | Example / Specification |
|---|---|---|
| FDA-Accepted T1D Simulator | Provides a validated in-silico cohort for initial algorithm development and safety testing. | UVA/Padova Simulator, OhioT1DM Simulator. |
| Real-World Dataset | For training and validating data-augmented models on noisy, complex physiological signals. | OhioT1DM Dataset (CGM, insulin, carbs). |
| Deep Learning Framework | Enables the construction and training of neural network components (LSTM, RNN). | TensorFlow, PyTorch. |
| Bayesian Optimization Library | Facilitates implementation of parameter personalization protocols. | GPyOpt, Scikit-Optimize, BoTorch. |
| Modeling & Simulation Environment | For implementing and integrating the differential equations of the Hovorka model. | MATLAB/Simulink, Python (SciPy, JAX). |
| Continuous Glucose Monitoring (CGM) Data Stream | Real-time glucose values for model input and error calculation. | Dexcom G6, Medtronic Guardian (Research interfaces). |
| Closed-Loop Research Platform | A safe sandbox to integrate the hybrid model into a full control-to-range algorithm. | AndroidAPS, OpenAPS loop cores, proprietary clinical platforms. |
Conclusion
The Hovorka model remains a cornerstone in the development of sophisticated closed-loop insulin delivery algorithms, offering a robust, physiologically grounded framework for glucose prediction and control. This guide has traversed its foundational principles, practical implementation methodologies, optimization for clinical variability, and rigorous validation benchmarks. For researchers and developers, the model provides a versatile platform, yet its efficacy is contingent upon meticulous personalization and integration with adaptive safety mechanisms. Future directions point toward the evolution of hybrid models that fuse this physiological paradigm with data-driven AI techniques, enhancing adaptability to complex daily life factors. Ultimately, the continued refinement and validation of Hovorka-based algorithms are pivotal for advancing the artificial pancreas from a research prototype to a widely accessible, personalized therapeutic device, promising improved quality of life and clinical outcomes for people with diabetes. The model's legacy lies in its proven contribution to a pathway that is steadily closing the loop in diabetes care.